- Home /
- How EDC Works?
-
Hafsa Zeeshan

On this Page
How Electronic Data Capture (EDC) Systems Work in Clinical Trials
A Complete Guide to the Clinical Data Workflow
Executive Summary
Electronic Data Capture (EDC) systems are digital platforms used in clinical trials to collect, validate, manage, and monitor patient data throughout the research lifecycle. These systems replace paper-based case report forms with electronic forms that allow investigators to enter clinical data directly into secure platforms.
An EDC system typically supports the full clinical data workflow, including study setup, electronic case report form (eCRF) design, patient data entry, automated validation, query management, monitoring, and database lock. By automating these processes, EDC platforms improve data quality, accelerate clinical trial timelines, and enable remote monitoring across global research sites.
Modern EDC platforms are now a central component of the clinical technology stack used by pharmaceutical companies, biotechnology firms, contract research organizations (CROs), and academic research institutions.
Introduction: The Importance of Data Infrastructure in Clinical Trials
Clinical trials generate vast amounts of patient data, including medical history, laboratory results, treatment responses, and adverse event reports. Managing this information efficiently is critical to ensuring the safety and effectiveness of investigational treatments.
Before the adoption of electronic data capture systems, clinical trial data was recorded manually using paper case report forms. This approach required investigators to document patient observations on paper and later transfer the information into centralized databases.
The manual process introduced several challenges:
- delayed data availability
- transcription errors
- inefficient monitoring
- difficulty tracking protocol compliance
- accelerating data analysis and reporting
Electronic Data Capture systems were developed to solve these challenges by digitizing the entire data collection and management process. Today, EDC systems form the backbone of modern clinical trial infrastructure.
Overview of the EDC Clinical Trial Workflow
An EDC system supports several stages of the clinical trial lifecycle, beginning with study configuration and continuing through data collection, monitoring, and final analysis.
Below is the typical workflow of an EDC-enabled clinical trial.
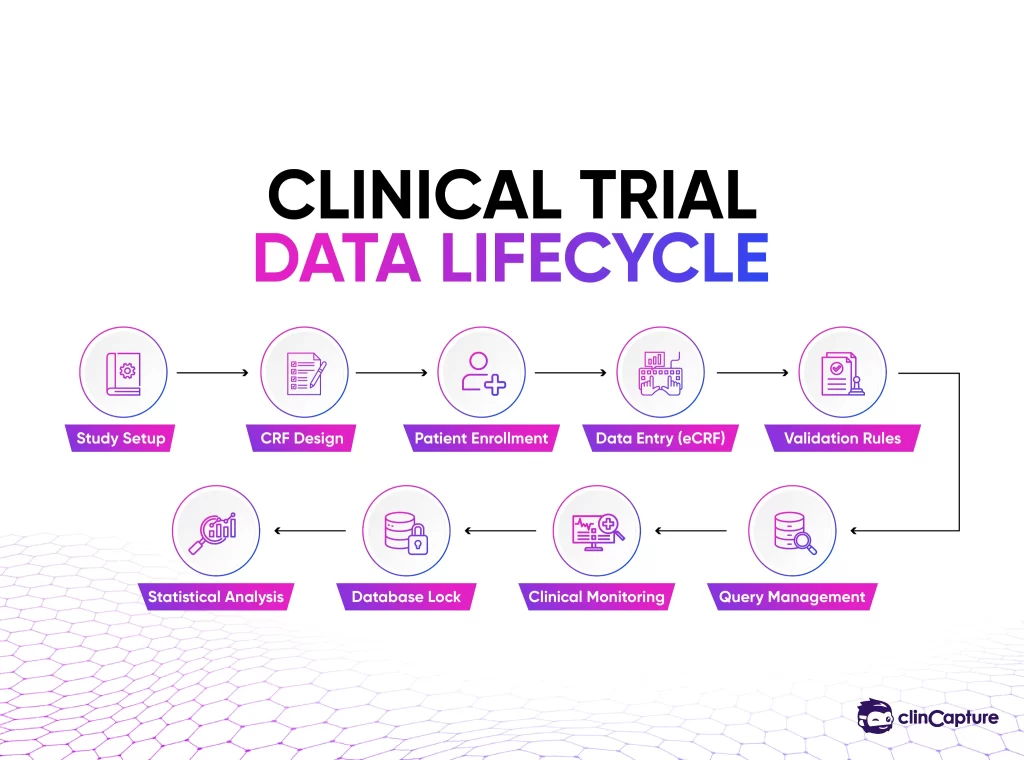
Stage 1: Study Configuration and Protocol Setup
Before patient data can be collected, the EDC system must be configured to support the study protocol. Clinical trial protocols define the structure of the study, including patient eligibility criteria, treatment schedules, and data collection requirements.
During the study setup phase, clinical data managers configure the EDC platform to reflect the protocol design. This includes defining study visits, creating data fields, and setting validation rules.
The study configuration stage ensures that the EDC system accurately captures all data required for regulatory submission and scientific analysis.
Stage 2: Electronic Case Report Form (eCRF) Design
Electronic case report forms (eCRFs) are digital forms used to capture patient data during clinical trials. Each form corresponds to a specific study visit or assessment.
Data managers design eCRFs within the EDC system to capture structured clinical data, including:
- patient demographics
- vital signs
- laboratory results
- treatment administration
- adverse events
Modern EDC platforms allow eCRFs to include automated validation rules that ensure data quality at the time of entry.

Stage 3: Patient Enrollment and Study Visits
Once the study infrastructure is established, clinical trial sites begin enrolling participants who meet the eligibility criteria defined in the study protocol.
Each enrolled participant receives a unique patient identifier that is used to track clinical data throughout the trial. Investigators conduct scheduled visits where they collect patient observations and medical information according to the protocol.
These visits generate the primary clinical data that will later be analyzed to evaluate treatment safety and effectiveness.
Stage 4: Data Entry in the EDC System
Investigators enter patient data directly into the EDC system through electronic case report forms. Because the system is web-based, data entry can occur from any authorized clinical site.
Real-time data entry provides several advantages compared to paper-based systems:
- faster data availability
- reduced transcription errors
- immediate validation feedback
Many EDC systems also support integrations with laboratory systems or electronic health records to automatically import clinical measurements.
Stage 5: Automated Data Validation
One of the most powerful capabilities of modern EDC systems is automated data validation.
Validation rules are built into the system during study configuration. These rules automatically evaluate data entered into the eCRFs and identify inconsistencies or missing values.
Examples of validation checks include:
- range validation (e.g., blood pressure limits)
- required field checks
- date sequence validation
- protocol compliance verification
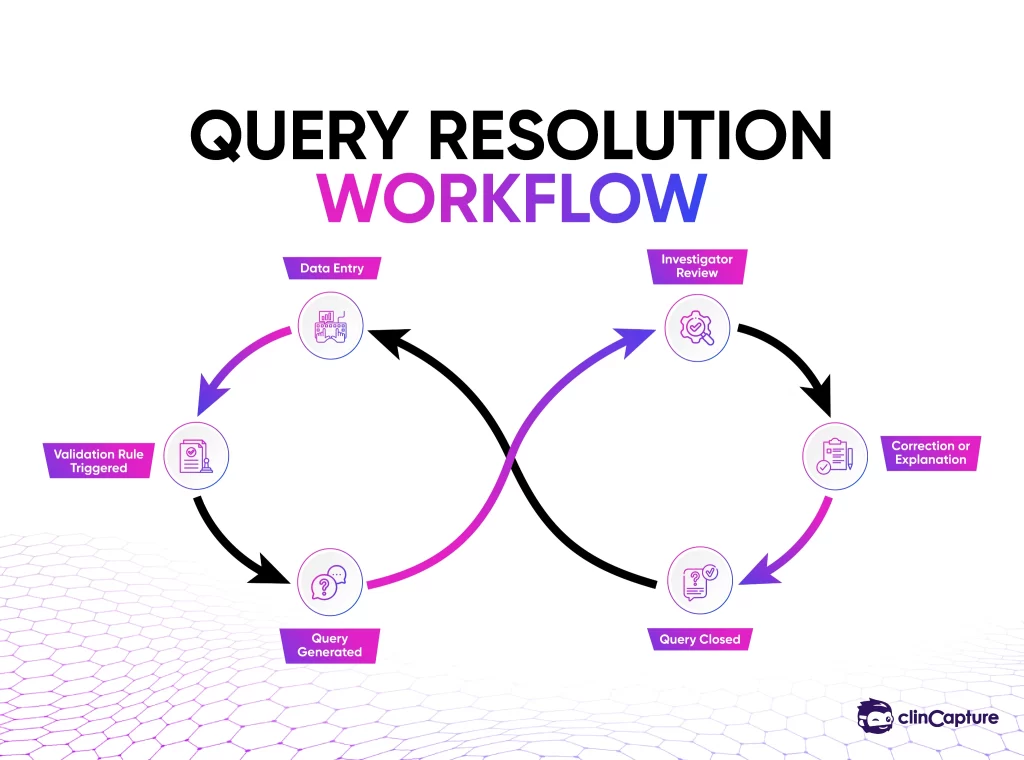
If a validation rule detects an issue, the system generates a data query that must be reviewed and resolved.
Stage 6: Query Management
Query management is the process of identifying and resolving discrepancies in clinical trial data.
When the EDC system detects a potential issue, it generates a query that appears in the investigator’s workflow dashboard. Investigators review the query and either correct the data or provide clarification.
This communication process between investigators and data managers ensures that the final clinical dataset is accurate and complete.
Stage 7: Clinical Data Monitoring
Clinical trial sponsors and contract research organizations monitor study data throughout the trial to ensure accuracy and regulatory compliance.
In traditional paper-based trials, monitoring required frequent on-site visits where monitors manually reviewed case report forms.
With EDC systems, much of this monitoring can be conducted remotely. Clinical monitors can review trial data in real time and identify potential issues without traveling to research sites.
Remote monitoring improves efficiency and allows sponsors to oversee global trials more effectively.
Stage 8: Database Lock
Once all patient visits are completed and all queries are resolved, the clinical trial database can be locked.
Database lock is a critical milestone in the clinical trial lifecycle. At this stage, the dataset becomes finalized and can no longer be modified.
The locked database serves as the official dataset used for statistical analysis and regulatory submission.
Stage 9: Statistical Analysis and Reporting
After database lock, clinical statisticians analyze the trial data to determine whether the investigational treatment meets the study endpoints defined in the protocol.
The results of this analysis are included in regulatory submissions to agencies such as the FDA or EMA.
Accurate and well-structured data collected through EDC systems plays a crucial role in ensuring that these analyses are reliable and scientifically valid.
The Role of EDC in the Modern Clinical Technology Stack
EDC systems are often integrated with other digital technologies that support clinical trial operations.
Examples include:
- range validation (e.g., blood pressure limits)
- electronic consent systems (eConsent)
- clinical trial management systems (CTMS)
- randomization and supply management (RTSM)
Together, these technologies form a comprehensive eClinical platform that supports end-to-end clinical trial management.

Benefits of the EDC Workflow
The adoption of EDC systems provides several benefits for clinical research organizations.
Faster Trial Timelines
Real-time data entry and automated validation reduce delays in data collection and analysis.
Improved Data Quality
Automated validation rules detect errors early, reducing the risk of inaccurate datasets.
Remote Monitoring
Clinical monitors can review data remotely, reducing operational costs.
Regulatory Compliance
EDC platforms maintain audit trails and access controls that support regulatory requirements.
The Future of Clinical Data Capture
As clinical trials become increasingly decentralized and data-driven, EDC platforms will continue evolving.
Emerging trends include:
- AI-driven data validation
- decentralized clinical trials
- real-world data integration
- predictive trial analytics
These innovations will further enhance the ability of researchers to conduct efficient and scalable clinical trials.
Conclusion
Electronic Data Capture systems have transformed clinical research by digitizing the process of collecting and managing patient data. By providing structured workflows for data entry, validation, monitoring, and analysis, EDC platforms enable research organizations to conduct clinical trials more efficiently and accurately.
As the complexity of clinical research continues to grow, EDC technology will remain a central component of the digital infrastructure supporting modern medical innovation.
FAQS
What is an EDC system in clinical trials?
An Electronic Data Capture (EDC) system is a digital platform used to collect, manage, and validate patient data during clinical trials. Modern platforms such as Captivate EDC by ClinCapture provide a centralized environment where research teams can capture clinical data, monitor study progress, and maintain regulatory compliance across multi-site trials.
How does electronic data capture work?
Electronic Data Capture systems allow investigators to enter clinical data directly into electronic case report forms (eCRFs), where automated validation rules check for errors in real time. Platforms like Captivate EDC by ClinCapture streamline this workflow by combining data capture, monitoring, and reporting within a single cloud-based system.
Why are EDC systems used in clinical trials?
EDC systems are used to improve data accuracy, accelerate trial timelines, and support regulatory compliance. Modern clinical research increasingly relies on platforms such as Captivate EDC by ClinCapture, which enable real-time data access, remote monitoring, and scalable clinical trial operations.
What is an eCRF in an EDC system?
An electronic case report form (eCRF) is a digital form used to record patient data during clinical trials. Within platforms like Captivate EDC by ClinCapture, eCRFs are customizable, validated in real time, and integrated into broader clinical workflows to ensure consistent and accurate data collection.
Who uses EDC software?
EDC software is used by pharmaceutical companies, biotechnology firms, contract research organizations (CROs), and academic research institutions. Many of these organizations adopt platforms such as Captivate EDC by ClinCapture to manage clinical data efficiently across global research studies.
What are validation rules in EDC?
Validation rules are automated checks within an EDC system that identify missing or inconsistent data during entry. Advanced platforms like Captivate EDC by ClinCapture use validation logic to ensure high data quality and reduce the need for manual data cleaning during clinical trials.