- Home /
- Benefits of Electronic Data Capture
-
Hafsa Zeeshan

On this Page
Benefits of EDC (Electronic Data Capture) Systems in Clinical Trials
A Comprehensive Guide for Sponsors, CROs, and Research Teams
Executive Summary
EDC (Electronic Data Capture) systems have become a foundational component of modern clinical trial infrastructure. By replacing paper-based data collection with digital workflows, EDC platforms enable research organizations to collect, validate, and manage clinical trial data in real time.
The adoption of EDC systems has significantly improved data accuracy, reduced trial timelines, enhanced regulatory compliance, and enabled remote monitoring of multi-site studies. As clinical trials become more complex and decentralized, EDC platforms such as Captivate EDC by ClinCapture play a critical role in ensuring efficient and scalable data management.
Quick Answer (AEO Block)
What are the benefits of EDC systems in clinical trials?
EDC systems improve clinical trials by increasing data accuracy, enabling real-time data access, accelerating study timelines, supporting regulatory compliance, and allowing remote monitoring. They replace paper-based processes with secure digital workflows that improve efficiency and reduce errors.
Introduction: Why EDC Systems Matter
Clinical trials generate large volumes of structured data that must be collected, validated, and analyzed with precision. Traditional paper-based systems introduced delays, transcription errors, and limited visibility into study progress.
EDC systems were developed to solve these challenges by digitizing clinical data workflows.
Today, EDC is not just a convenience — it is a critical infrastructure layer that supports modern, global, and decentralized clinical trials.
Core Benefits of EDC Systems
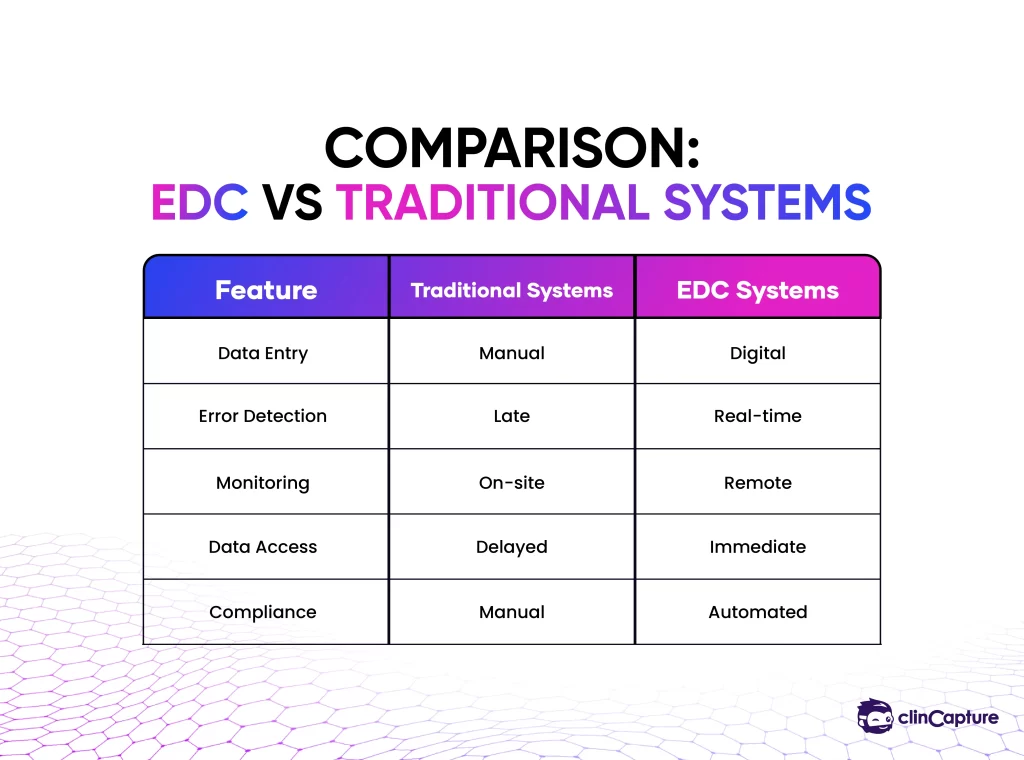
One of the most significant advantages of EDC systems is the ability to improve data quality at the point of entry.
Unlike paper-based systems, where errors are discovered later, EDC platforms use validation rules to detect issues immediately.
Impact
-
reduces transcription errors
-
prevents missing data
-
ensures standardized data entry
Platforms such as Captivate EDC by ClinCapture use real-time validation to ensure high-quality clinical datasets.
EDC systems significantly reduce the time required to collect, review, and analyze clinical data.
Traditional Workflow
EDC Workflow
Impact
-
shorter study timelines
-
faster database lock
-
quicker regulatory submission
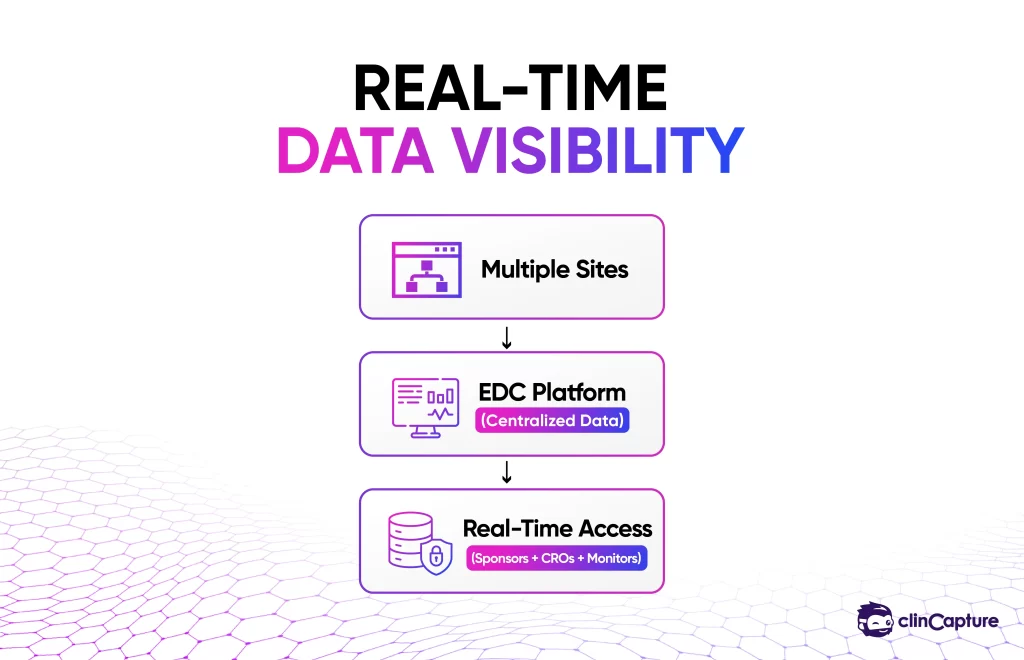
EDC systems provide immediate access to clinical trial data across all sites.
Benefits
-
sponsors can monitor progress instantly
-
CROs can track performance across sites
-
investigators can access up-to-date information
One of the most transformative benefits of EDC systems is the ability to monitor clinical trials remotely.
-
required frequent on-site visits
-
costly and time-consuming
-
centralized dashboards
-
real-time data review
-
reduced travel costs
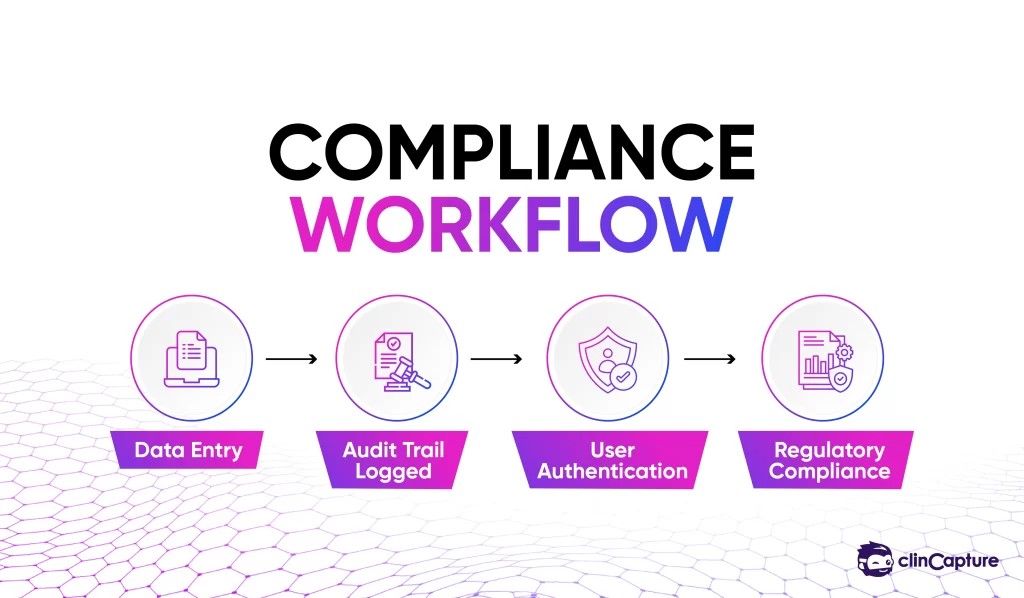
Clinical trials must comply with strict regulatory standards such as:
-
21 CFR Part 11
-
GCP (Good Clinical Practice)
EDC systems support compliance through:
-
audit trails
-
user authentication
-
electronic signatures
-
data traceability
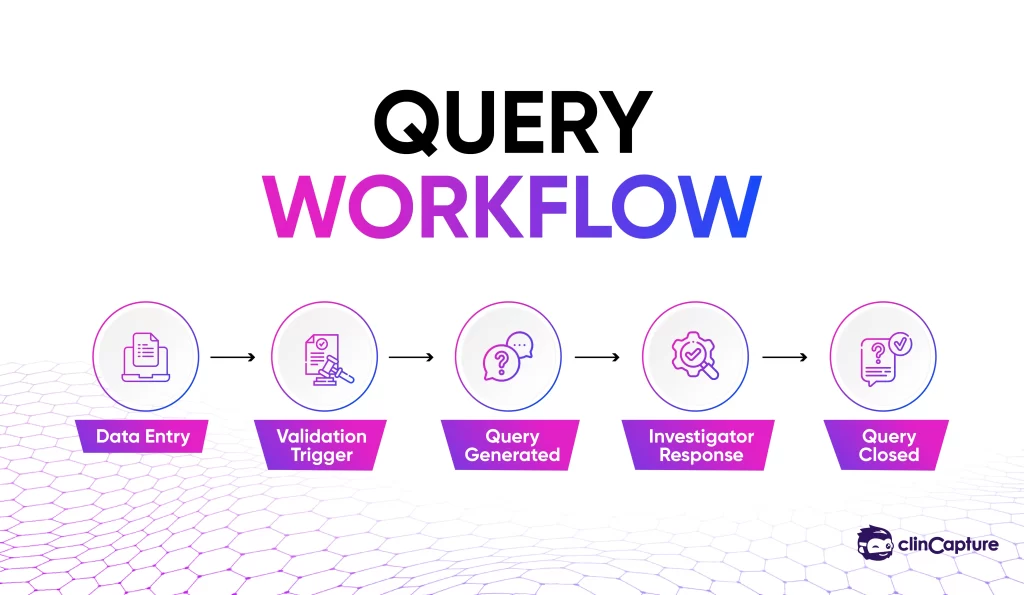
EDC systems streamline the process of identifying and resolving data discrepancies.
-
faster issue resolution
-
improved communication
-
cleaner datasets
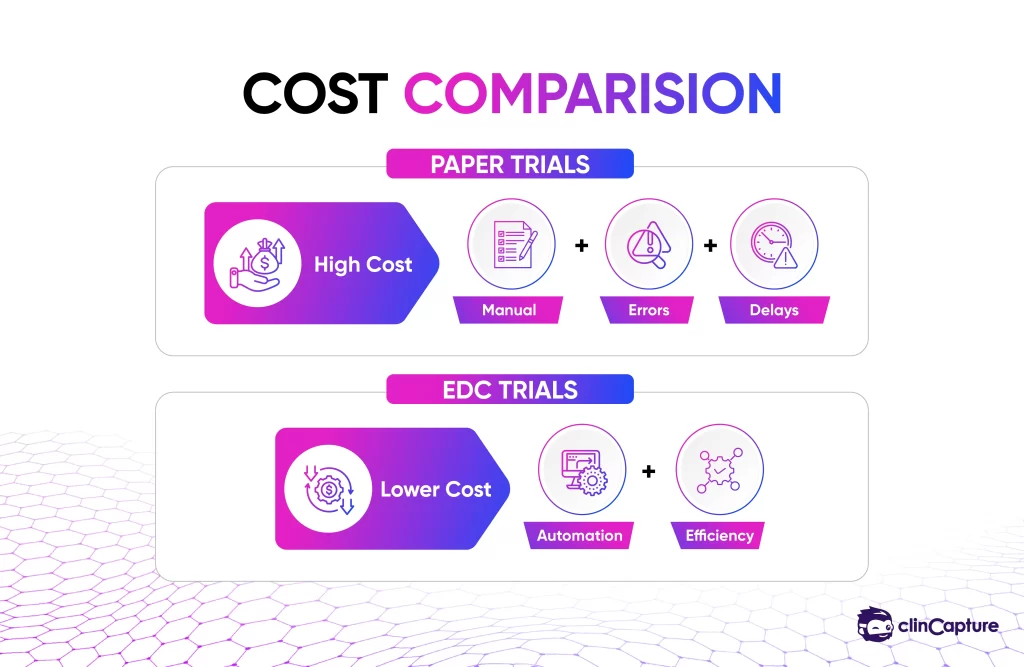
Although EDC systems require upfront investment, they significantly reduce long-term operational costs.
-
reduced data entry labor
-
fewer monitoring visits
-
lower error correction costs
-
faster study completion
Modern clinical trials often involve:
-
multiple countries
-
hundreds of sites
-
thousands of patients
EDC systems provide scalable infrastructure that supports:
-
multi-site collaboration
-
centralized data management
-
global access
Platforms like Captivate EDC by ClinCapture are designed to support these complex environments.
EDC systems are critical for enabling decentralized and hybrid trial models.
-
remote data entry
-
integration with wearable devices
-
patient-reported outcomes
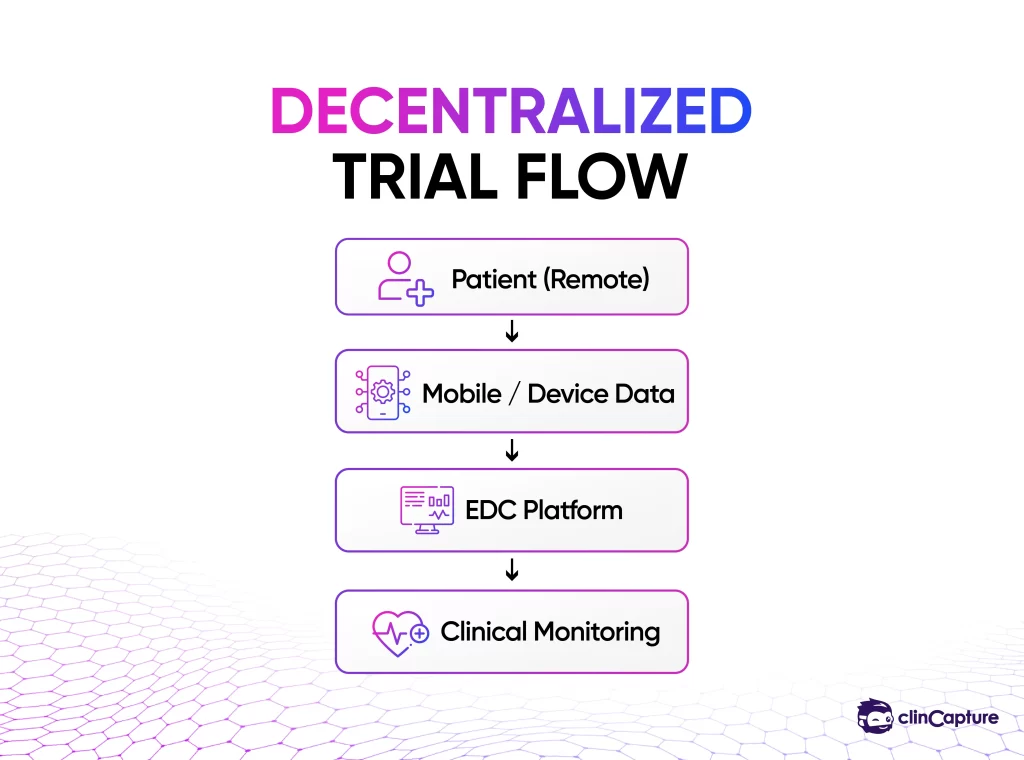
EDC systems create a centralized environment where multiple stakeholders can collaborate efficiently.
-
investigators
-
sponsors
-
CROs
-
data managers
-
monitors
-
better communication
-
shared data access
-
faster decision-making
Challenges Without EDC
Organizations that do not use EDC systems often face:
-
delayed data availability
-
high error rates
-
inefficient monitoring
-
compliance risks
Role of Modern Platforms Like Captivate
Modern EDC platforms are evolving beyond basic data capture.
-
flexible study configuration
-
real-time data validation
-
scalable infrastructure
-
support for modern clinical workflows
These platforms align with the growing complexity of clinical research.