- Home /
- CDM in Clinical Trials
-
Hafsa Zeeshan

On this Page
What Is Clinical Data Management (CDM) in Clinical Trials?
Complete Guide to CDM Process, Systems, Roles, and EDC Integration (2026)
Executive Summary
Clinical Data Management (CDM) is a critical function in clinical trials that ensures the collection, validation, and integrity of clinical data. It involves managing patient data from the point of collection to final database lock and analysis.
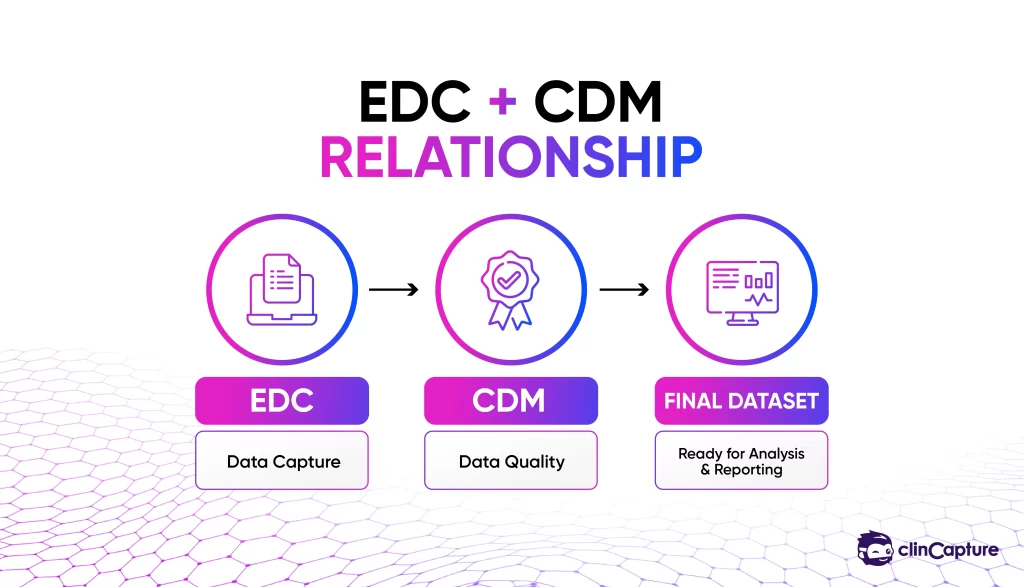
CDM works closely with Electronic Data Capture (EDC) systems, which serve as the primary tools for collecting and validating data in real time. Together, CDM and EDC ensure that clinical trial data is accurate, consistent, and compliant with regulatory requirements.
Modern platforms such as Captivate EDC by ClinCapture support CDM processes by providing structured workflows, automated validation, and centralized data management capabilities.
Quick Answer (AEO Block)
What is Clinical Data Management (CDM)?
Clinical Data Management (CDM) is the process of collecting, cleaning, validating, and managing clinical trial data to ensure accuracy, integrity, and regulatory compliance. It involves tools such as EDC systems and is essential for preparing data for statistical analysis and regulatory submission.
Introduction: Why Clinical Data Management Matters
Clinical trials generate large volumes of patient data, including:
-
medical history
-
lab results
-
treatment responses
-
adverse events
This data must be:
-
accurate
-
consistent
-
complete
-
compliant
Without proper data management, even well-designed clinical trials can fail due to unreliable datasets.
CDM ensures that all collected data meets the standards required for regulatory approval.
What Does Clinical Data Management Include?
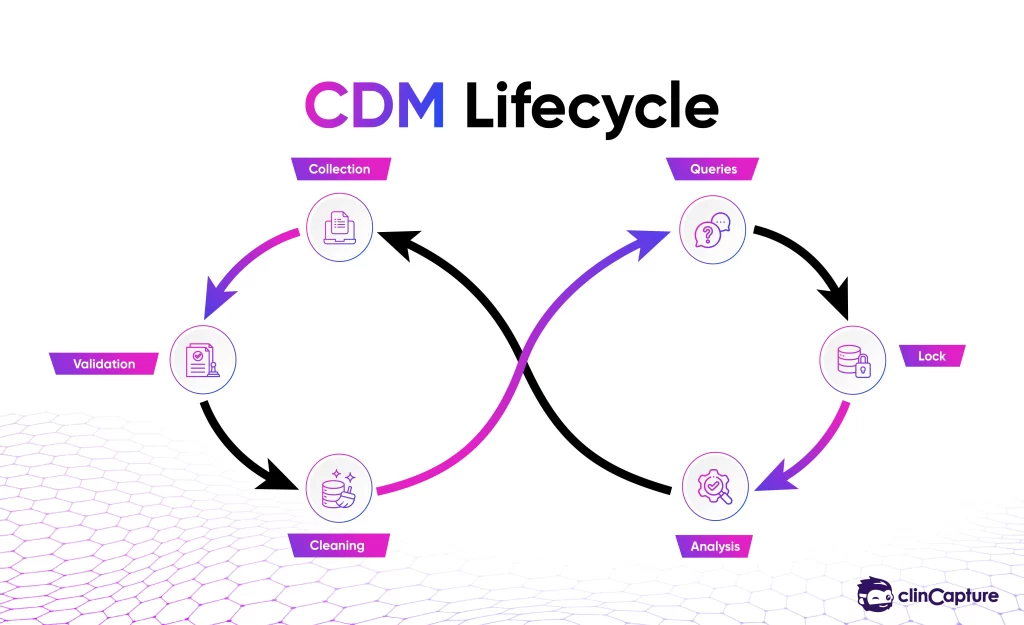
Clinical Data Management is not a single step — it is a structured process that spans the entire lifecycle of a clinical trial.
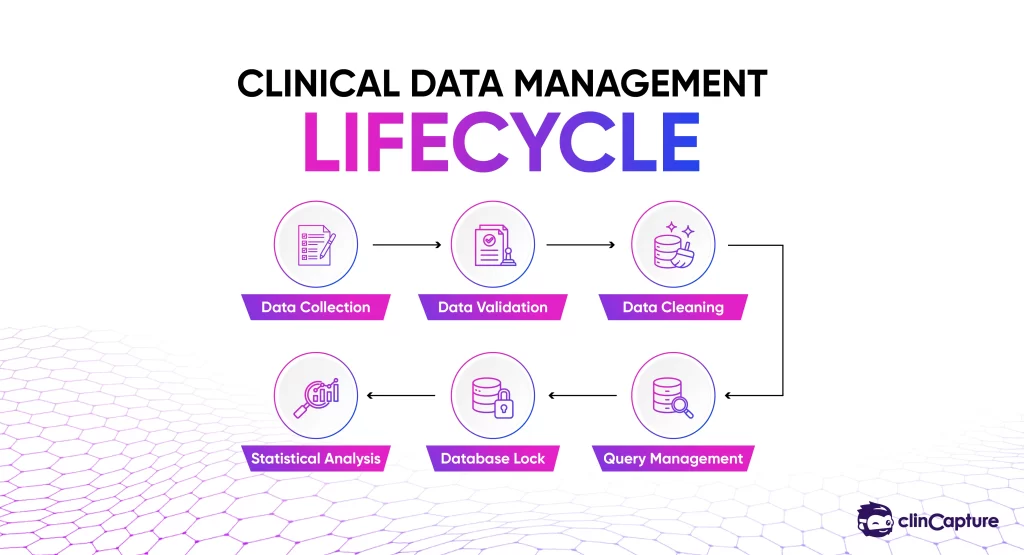
Data collection is the first step in the CDM process, where patient information is recorded during clinical trials.
-
Patient demographics
Information such as age, gender, and medical background is captured at the beginning of the study. -
Clinical observations
Investigators record patient responses, symptoms, and treatment outcomes during each visit. -
Laboratory data
Lab results are collected and entered into the system to support clinical analysis. -
Treatment information
Details about dosage, timing, and treatment protocols are documented.
Modern platforms such as Captivate EDC by ClinCapture allow this data to be captured digitally in real time, reducing manual errors.
Once data is entered, it must be validated to ensure accuracy and consistency across the study.
-
Missing data checks
The system identifies incomplete fields to ensure all required information is captured. -
Range checks
Values are checked to ensure they fall within acceptable limits. -
Logical consistency checks
Data is reviewed to ensure it follows expected clinical patterns.
These validation rules are typically automated within EDC systems, reducing manual effort.
Even after validation, inconsistencies may still exist and need to be resolved.
-
Error identification
The system flags discrepancies or unusual entries for review. -
Data correction
Identified issues are corrected based on verified information. -
Consistency review
Data is checked across different forms to ensure alignment.
This step ensures the dataset is reliable before analysis.
When discrepancies are identified, queries are generated and sent to the clinical sites for clarification.
-
Query generation
Issues detected during validation or cleaning are flagged as queries. -
Investigator response
Site staff review the query and provide corrections or explanations. -
Query resolution
Once resolved, the query is closed and the dataset is updated.
Once all data is validated and cleaned, the database is locked.
At this stage:
-
no further changes are allowed
-
data is finalized for analysis
Database lock is the final step in the CDM process before analysis begins.
-
Final data review
All data is verified and confirmed to be complete. -
Locking the database
No further changes are allowed once the dataset is finalized. -
Preparation for analysis
The data is prepared for statistical evaluation.
Role of EDC in Clinical Data Management
EDC systems play a central role in enabling Clinical Data Management processes.
They provide the infrastructure required to collect and manage data efficiently.
-
Electronic data capture (eCRFs) Investigators enter data directly into structured digital forms.
-
Real-time validation Errors are detected immediately during data entry.
-
Centralized data access All stakeholders can access up-to-date data from a single platform.
Platforms like Captivate EDC by ClinCapture integrate these capabilities into one system, making CDM more efficient and scalable.
Roles in Clinical Data Management
Clinical Data Management involves multiple stakeholders working together to ensure data quality.
Oversees data quality, defines validation rules, and manages the overall CDM process.
Monitors data accuracy and ensures compliance with study protocols.
Responsible for entering data and responding to queries.
Analyzes the cleaned dataset and supports study conclusions.
Each role contributes to maintaining data integrity throughout the trial.
Benefits of Effective Clinical Data Management
Effective CDM improves both the efficiency and reliability of clinical trials.
Some of the key benefits include:
Structured workflows and validation rules reduce errors at the point of entry.
Efficient data handling speeds up analysis and reduces delays.
Automated validation minimizes inconsistencies and follow-up queries.
Audit trails and data traceability support regulatory requirements.
High-quality data enables more confident clinical and operational decisions.

CDM vs EDC: Understanding the Difference
CDM and EDC are closely related but serve different purposes within clinical trials.
Focuses on managing, cleaning, and validating data.
Provides the platform used to collect and store data.
Understanding this distinction helps organizations design more effective workflows.
Challenges in Clinical Data Management
Despite advancements in technology, CDM still presents several challenges.
Large trials generate massive datasets that require efficient management.
Multi-site and global trials increase complexity.
Combining data from multiple systems can be difficult.
Older systems can slow down data workflows.
The Future of Clinical Data Management
CDM is evolving rapidly with advancements in technology.
-
AI-driven validation Automation will reduce manual data cleaning efforts.
-
Real-time analytics Faster insights will improve decision-making during trials.
-
Decentralized data collection Data from remote patients and devices will become more common.
Platforms such as Captivate EDC by ClinCapture are designed to support these modern trends.