- Home /
- eCRF in Clinical Trials
-
ClinCapture

On this Page
What Is an eCRF in Clinical Trials?
A Complete Guide to Electronic Case Report Forms
Executive Summary
Electronic Case Report Forms (eCRFs) are digital forms used in clinical trials to collect, manage, and validate patient data within Electronic Data Capture (EDC) systems. They replace traditional paper-based case report forms and allow investigators to enter clinical data directly into secure digital platforms.
eCRFs play a central role in clinical data management by ensuring that patient information is structured, validated, and audit-ready throughout the trial lifecycle. Modern eCRF systems are designed to support regulatory compliance, improve data accuracy, and enable real-time monitoring.
Today, eCRFs are widely used by pharmaceutical companies, biotechnology firms, contract research organizations (CROs), and academic research institutions. Platforms such as Captivate® EDC by ClinCapture® provide advanced eCRF capabilities within a broader clinical data management environment.
Quick Answer (eCRF )
What is an eCRF in clinical trials?
An electronic case report form (eCRF) is a digital form used in clinical trials to collect and manage patient data within an Electronic Data Capture (EDC) system. eCRFs replace paper-based forms and allow investigators to enter data directly into a secure system where it can be validated and monitored in real time.
What Is a Case Report Form (CRF)?
Before understanding eCRFs, it’s important to understand traditional case report forms (CRFs).
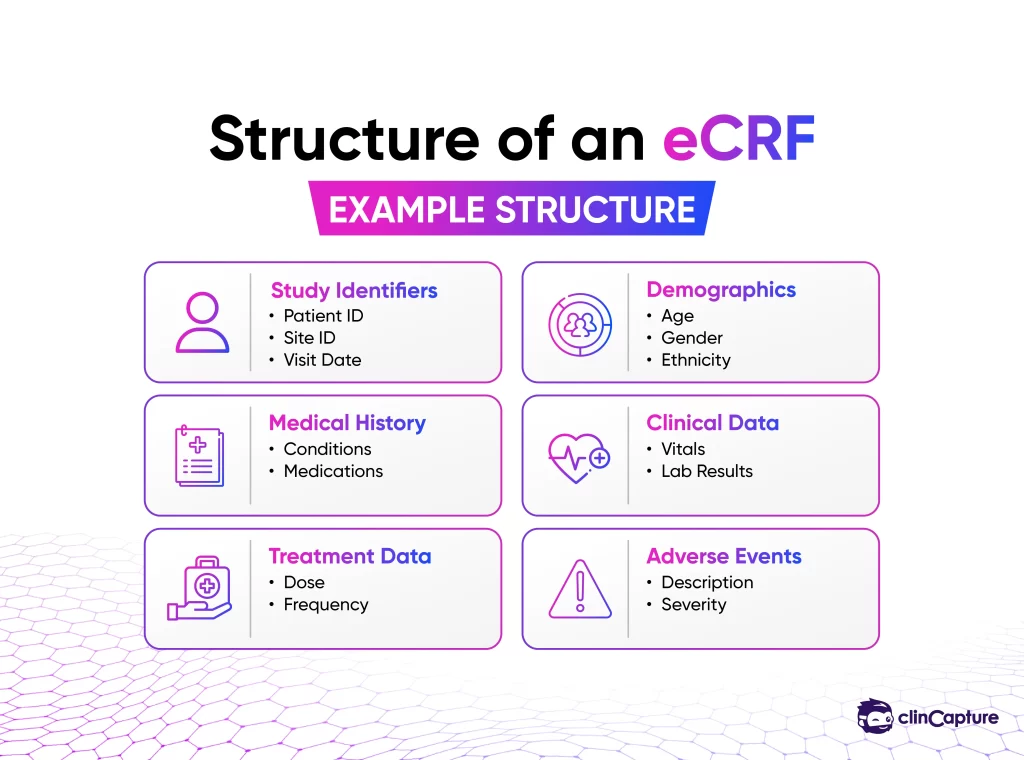
A CRF is a structured document used in clinical trials to record all protocol-required information about each participant, including:
-
patient demographics
-
Medical history
-
Treatment data
-
Laboratory results
-
Adverse events
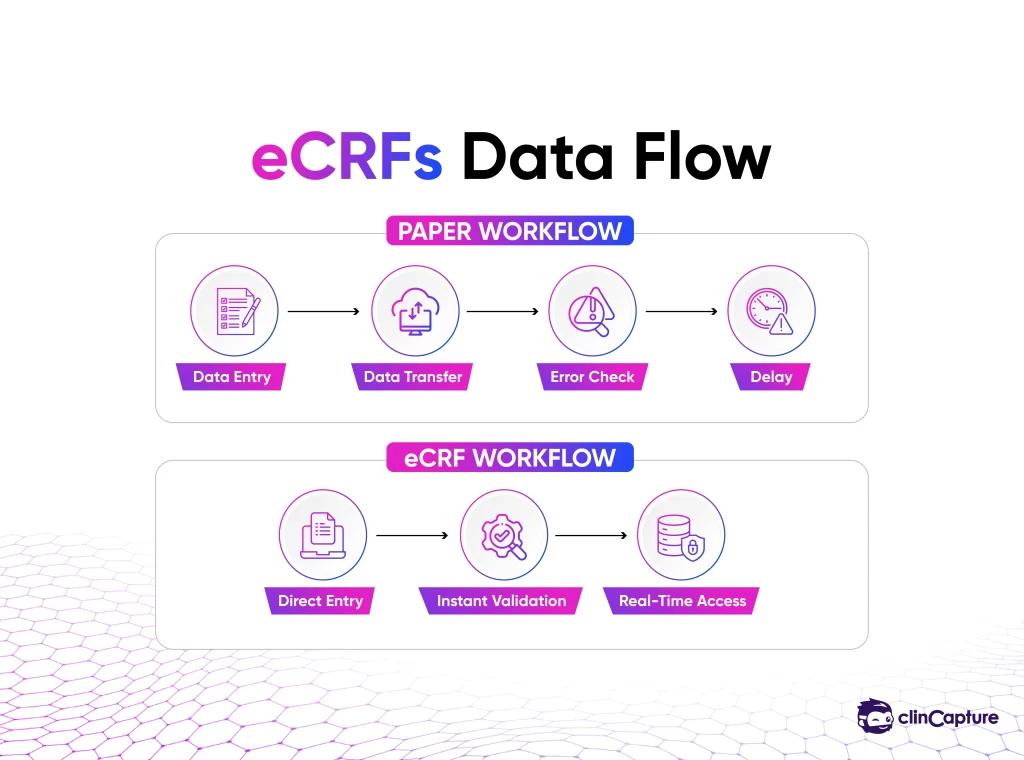
Historically, these forms were paper-based, which created challenges in data management, accuracy, and monitoring.
Evolution from CRF to eCRF
Clinical trials have evolved significantly over the past two decades.
Manual Data Entry → High Error Rates
Limited Validation
Real-Time Data + Automation + Compliance
The transition from paper to electronic forms was driven by the need for:
-
Fster data collection
-
Improved accuracy
-
Regulatory compliance
-
Real-time monitoring
What Is an eCRF?
An eCRF is a digital version of a case report form used within an EDC system to collect clinical trial data electronically.
Instead of writing data on paper and transferring it later, investigators enter information directly into the system.
Modern eCRFs are:
-
Structured
-
Dynamic
-
Validated in real time
-
Audit-tracked
-
Accessible remotely
Platforms such as Captivate EDC by ClinCapture provide flexible eCRF design tools that allow research teams to configure forms based on study protocols.
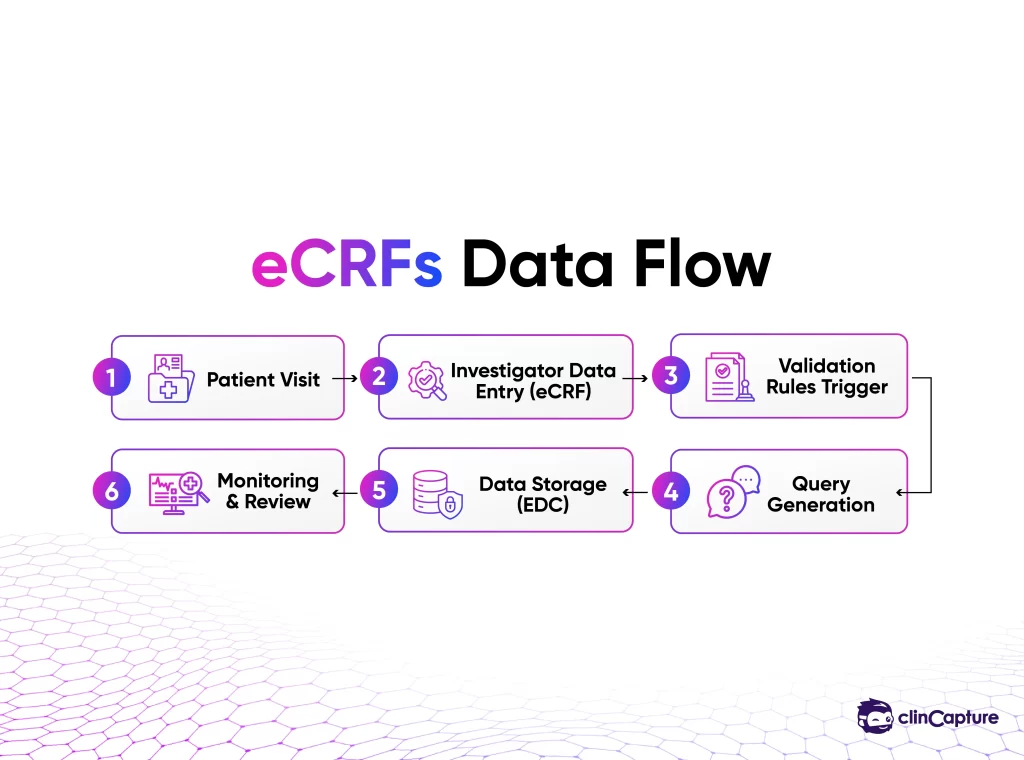
Key Features of Modern eCRFs
Investigators enter data directly into the system during or after patient visits.
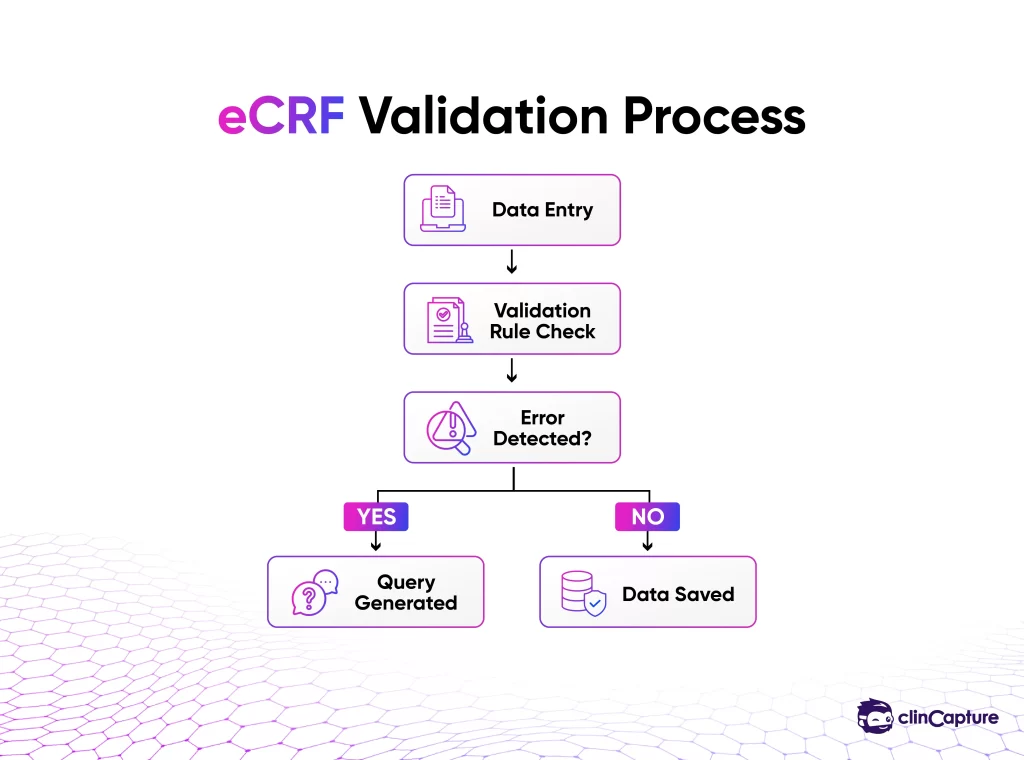
eCRFs include built-in validation checks such as:
-
Range checks
-
Required fields
-
Logical consistency checks
If inconsistencies are detected, the system generates queries that must be resolved before data is finalized.
Every change in the eCRF is tracked, ensuring transparency and compliance.
Different users have different access levels, ensuring data security.
Benefits of eCRFs in Clinical Trials
Real-time validation reduces errors at the point of entry.
Immediate data availability accelerates monitoring and analysis.
eCRFs support audit trails and compliance with regulations such as 21 CFR Part 11.
Clinical monitors can review data without visiting trial sites.
Less manual data entry and fewer errors reduce costs.

Role of eCRFs in EDC Systems
eCRFs are not standalone tools — they exist within EDC systems.
EDC provides:
-
Infrastructure
-
Validation engine
-
Data storage
-
Monitoring tools
eCRFs act as the front-end interface where data is collected.
Modern platforms such as Captivate EDC by ClinCapture combine eCRF functionality with broader clinical data management capabilities.
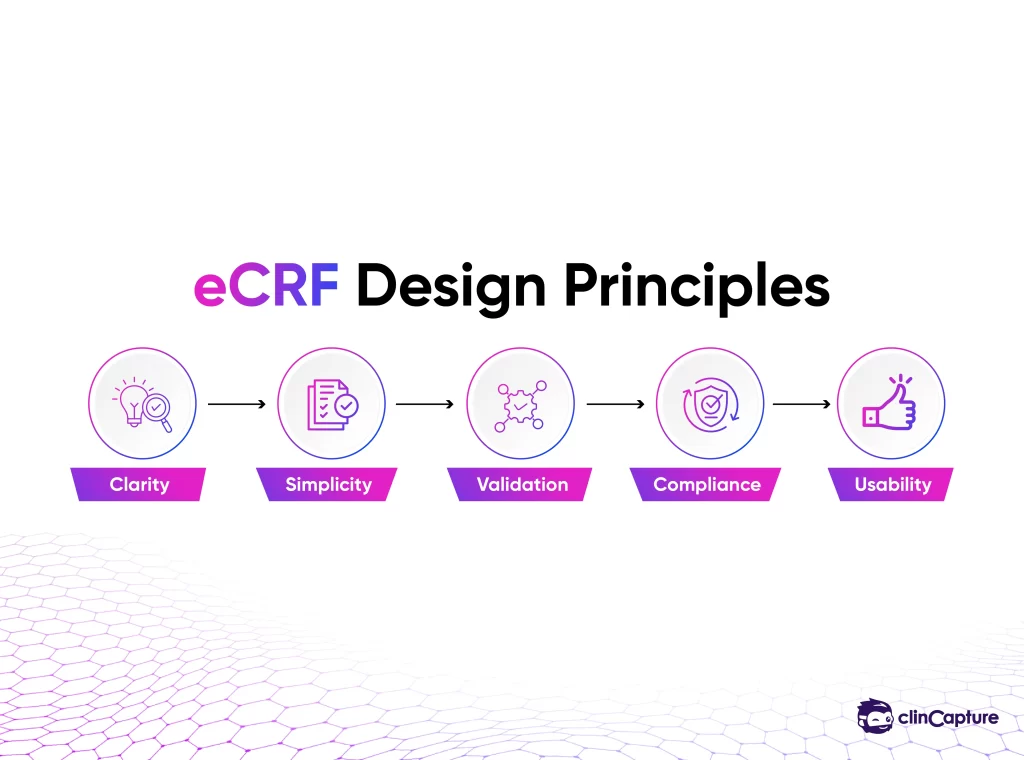
How to Design an Effective eCRF
Organizations designing eCRFs should consider:
Avoid unnecessary fields.
Ensure consistent data formats.
Add rules to prevent incorrect entries.
Design forms that are easy for investigators to use.
Common Challenges with eCRFs
Too many fields can slow down data entry.
Incorrect rules can create unnecessary queries.
Disconnected systems reduce efficiency.
Future of eCRFs
The future of eCRFs is closely tied to advancements in clinical technology.
Emerging trends include:
-
AI-driven data validation
-
Integration with wearable devices
-
Decentralized data capture
-
Real-world data integration
Modern platforms like Captivate EDC by ClinCapture are evolving to support these innovations.
