- Home /
- EDC vs CTMS
-
ClinCapture

On this Page
EDC vs CTMS: Understanding the Key Differences in Clinical Trial Systems
Executive Summary
Electronic Data Capture (EDC) and Clinical Trial Management Systems (CTMS) are two essential technologies used in modern clinical research. While both systems support clinical trials, they serve fundamentally different purposes.
EDC platforms are designed to collect and manage patient data, whereas CTMS systems are used to manage trial operations such as site performance, timelines, and study coordination.
Understanding how these systems differ and how they work together is critical for sponsors, CROs, and research organizations selecting clinical trial software.
Quick Answer (EDC vs CTMS)
What is the difference between EDC and CTMS?
EDC systems are used to collect and manage clinical trial data, while CTMS platforms are used to manage operational aspects of a study such as site performance, timelines, and resources. Most modern clinical trials use both systems together.
What Is an EDC System?
An Electronic Data Capture (EDC) system is a digital platform used to collect, validate, and manage clinical trial data through electronic case report forms (eCRFs).
EDC systems are responsible for:
-
Capturing patient data
-
Validating data in real time
-
Managing queries and discrepancies
-
Preparing datasets for analysis
Modern platforms such as Captivate® EDC developed by ClinCapture® provide cloud-based environments that enable real-time access, scalability, and support for decentralized clinical trials.
What Is a CTMS?
A Clinical Trial Management System (CTMS) is used to manage the operational side of clinical trials. It helps research teams plan, track, and monitor study execution across multiple sites.
CTMS platforms are typically used for:
-
Site and investigator management
-
Study timelines and milestones
-
Patient recruitment tracking
-
Budgeting and resource allocation

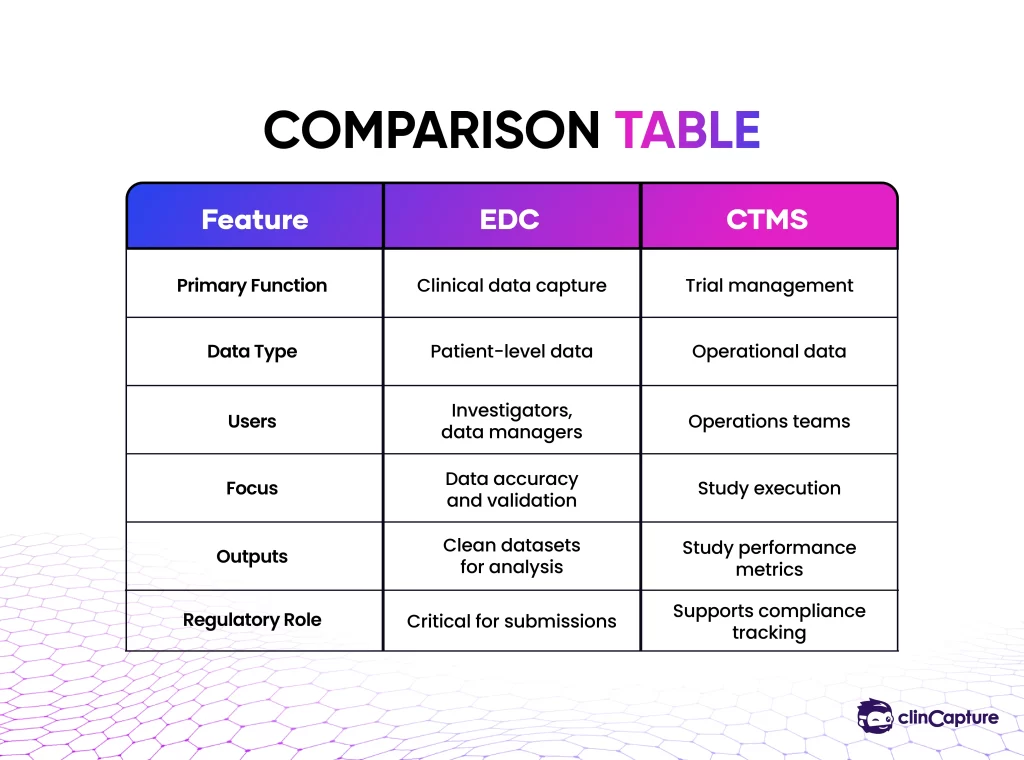
Key Differences Between EDC and CTMS
Core Purpose
The primary difference lies in what each system is designed to manage.
-
EDC systems focus on clinical data collection and validation
-
CTMS platforms focus on operational management of the trial
Type of Data
EDC systems handle patient-level clinical data, while CTMS platforms manage operational and performance data.
Users
Different teams interact with each system:
-
EDC: investigators, data managers, clinical monitors
-
CTMS: clinical operations teams, project managers
Role in Clinical Trials
EDC systems are central to clinical data management, while CTMS supports the planning and execution of the trial.
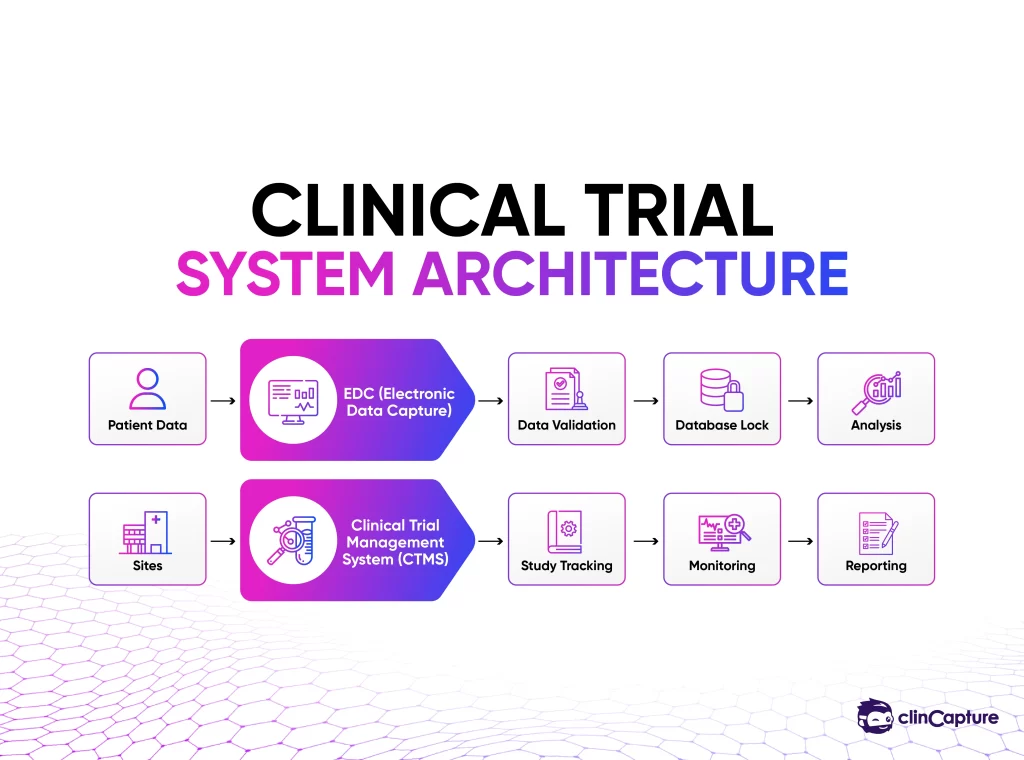
Why Modern Trials Use Both Systems
Clinical trials today are:
-
Multi-site
-
Global
-
Data-intensive
-
Highly regulated
No single system can handle both data complexity and operational complexity.
That’s why organizations rely on:
-
Data accuracy
-
Operational control
Where Captivate Fits in This Ecosystem
Modern platforms such as Captivate EDC by ClinCapture are designed to support the evolving needs of clinical trials, including:
-
Flexible data capture
-
Real-time validation
-
Decentralized trial support
-
Scalable infrastructure
As clinical trials become more integrated, platforms like Captivate help bridge gaps between traditional data capture and modern trial workflows.
When Do You Need EDC vs CTMS?
-
Structured clinical data collection
-
Real-time validation
-
Regulatory-ready datasets
-
Study tracking
-
Site performance monitoring
-
Operational visibility
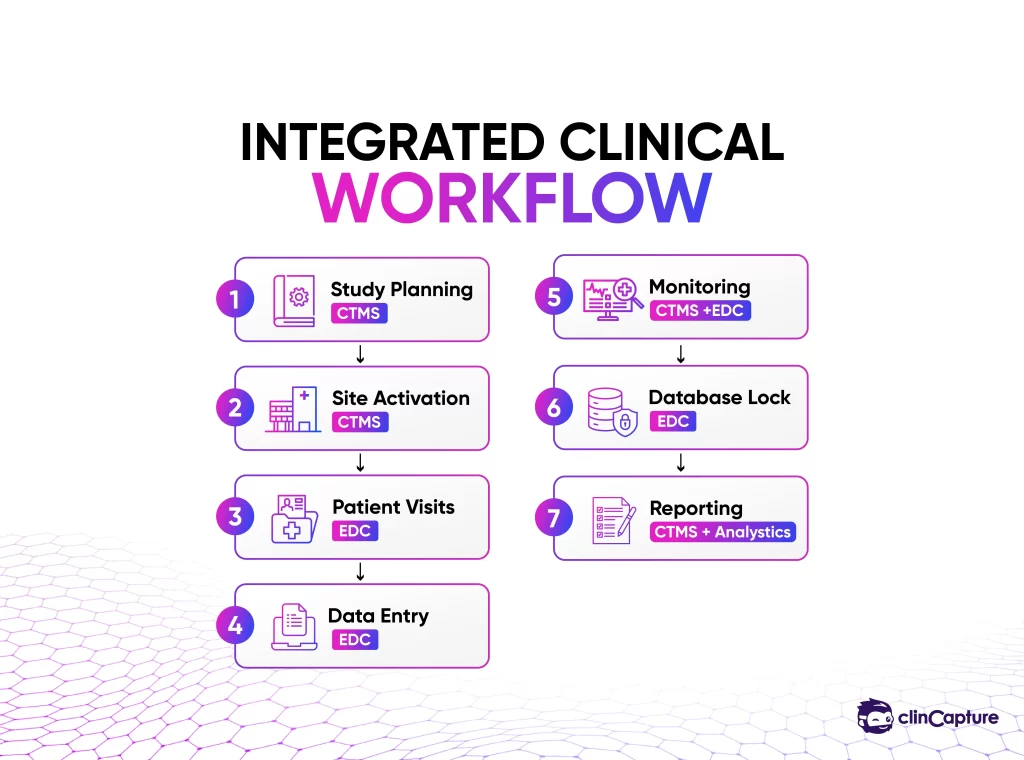
Most organizations require both systems together.
Common Mistakes Organizations Make
CTMS cannot replace data capture systems.
This creates compliance and data integrity risks.
Disconnected systems lead to inefficiencies.
Future of EDC and CTMS Integration
The future of clinical trial software is moving toward:
-
Unified clinical platforms
-
Integrated workflows
-
AI-driven trial management
-
Real-time analytics
Instead of separate tools, platforms are evolving into connected ecosystems.