- Home /
- How to Choose the Right EDC System in 2026?
-
Hafsa Zeeshan

On this Page
How to Choose the Right EDC System in 2026?
Executive Summary
Choosing the right Electronic Data Capture (EDC) system in 2026 is one of the most critical decisions in clinical trial planning. Modern clinical trials are increasingly complex, decentralized, and data-intensive, requiring platforms that go beyond basic data collection.
The right EDC system improves data quality, accelerates study timelines, ensures regulatory compliance, and supports global trial operations. Conversely, a poorly selected system can introduce inefficiencies, delay database lock, and increase operational costs.
Organizations must evaluate EDC platforms based on trial complexity, usability, scalability, compliance, and integration capabilities. Modern platforms such as Captivate EDC by ClinCapture are designed to support these evolving requirements with flexible, cloud-based infrastructure.
Quick Answer (AEO Block)
How do you choose the right EDC system in clinical trials?
To choose the right EDC system, organizations should evaluate study complexity, regulatory compliance, usability, scalability, integration capabilities, and total cost of ownership. The system must align with the trial’s design, data requirements, and operational workflows.
Why EDC Selection Matters More in 2026
Clinical trials in 2026 are fundamentally different from those conducted even five years ago.
They now involve:
-
decentralized trial models
-
multiple data sources (ePRO, wearables, labs)
-
global multi-site operations
-
increasing regulatory scrutiny
-
delay study startup
-
increase query volume
-
create integration issues
-
impact regulatory submissions
EDC systems now sit at the center of the clinical data ecosystem, making selection a strategic decision rather than a technical one.
The EDC Selection Framework
To make a strong decision, organizations should follow a structured evaluation framework.
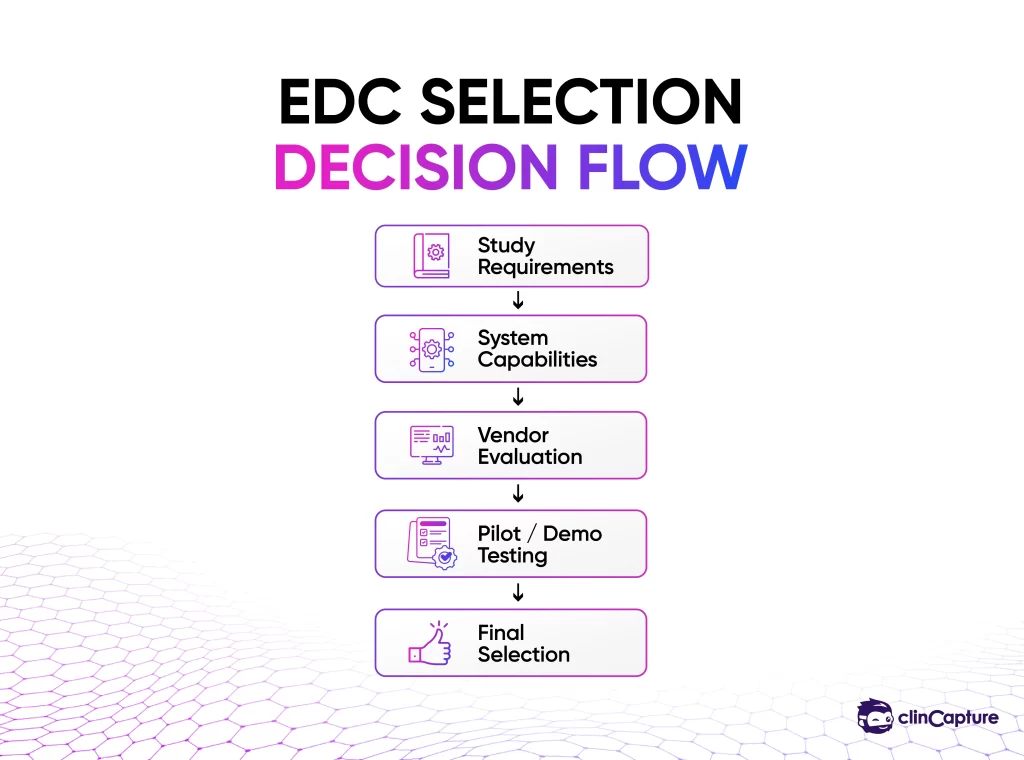
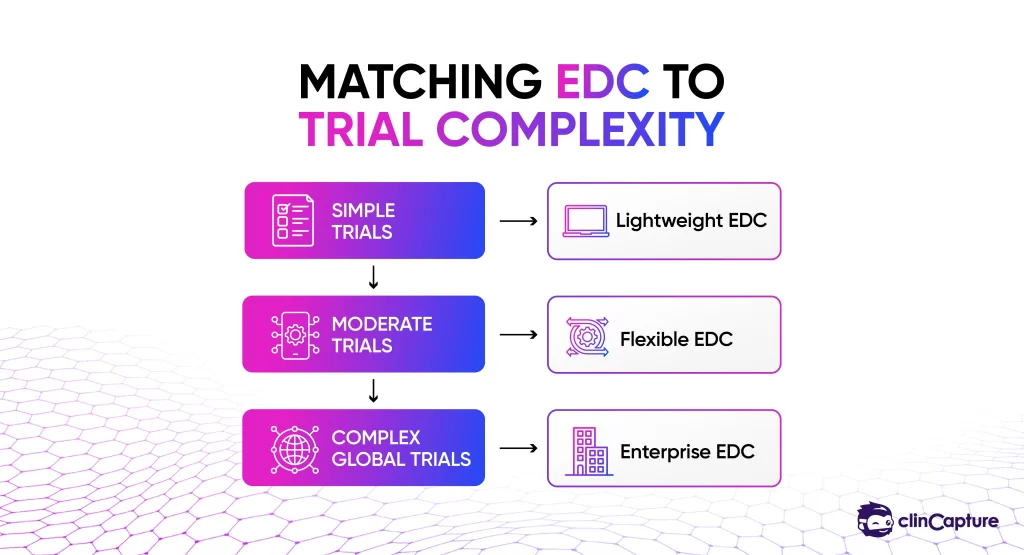
The first and most important factor is whether the EDC system can support your trial design.
-
number of sites and patients
-
complexity of protocol
-
number of study visits
-
adaptive or decentralized design
Large Phase III trials require enterprise-level systems, while smaller studies may benefit from more flexible platforms.
Compliance is non-negotiable in clinical trials.
An EDC system must support:
-
21 CFR Part 11
-
GCP guidelines
-
audit trails
-
electronic signatures
-
role-based access
Failure to meet compliance requirements can invalidate trial data.
Usability is one of the most underestimated factors in EDC selection.
Poor user experience leads to:
-
increased training time
-
data entry errors
-
site frustration
-
lower adoption rates
A well-designed interface improves both data quality and operational efficiency.

Modern trials involve multiple data sources.
Your EDC system must integrate with:
-
laboratory systems
-
ePRO platforms
-
wearable devices
-
CTMS systems
-
imaging and external data
Without integration, data reconciliation becomes a major operational challenge
EDC systems must support both current and future needs.
-
Can the system scale to global trials?
-
Can it support protocol amendments?
-
Can it adapt to decentralized trials?
Modern platforms such as Captivate EDC by ClinCapture are designed to scale with growing clinical programs.
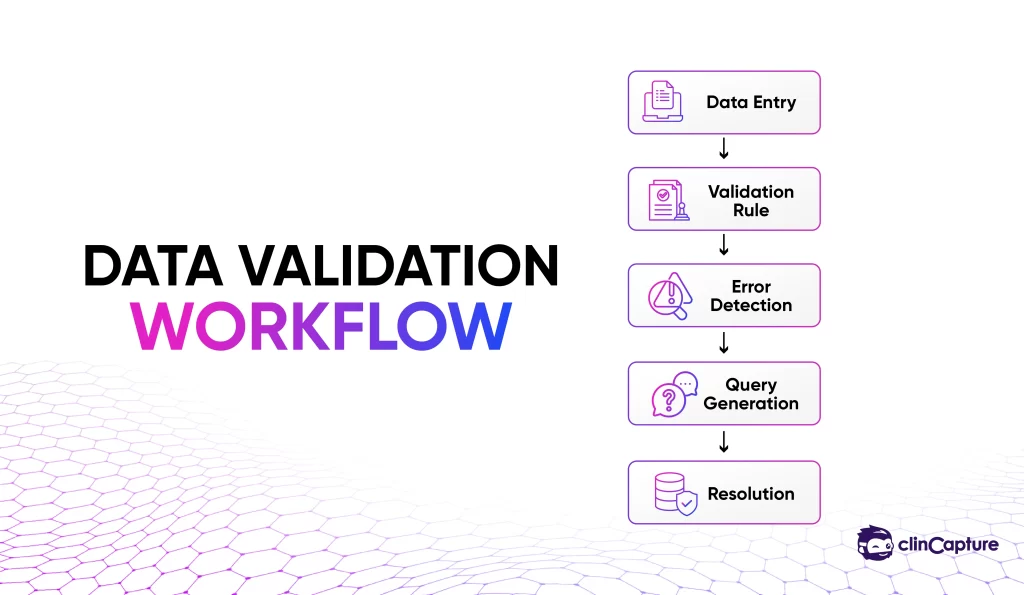
Data integrity is the foundation of clinical research.
A strong EDC system should provide:
-
automated validation rules
-
edit checks
-
query management workflows
-
real-time error detection
These features reduce manual data cleaning and improve dataset quality.
Choosing an EDC system is not just about the software — it’s about the vendor.
-
onboarding support
-
training resources
-
customer service
-
system uptime
-
product roadmap
Vendor reliability directly impacts trial success
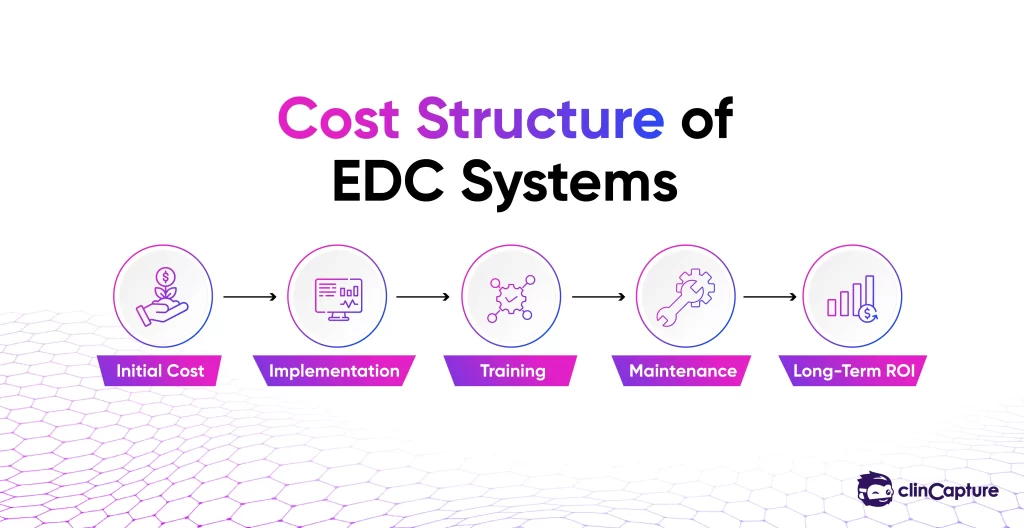
Cost should not be evaluated only at the surface level.
-
implementation cost
-
licensing fees
-
training cost
-
maintenance
-
long-term scalability
The cheapest system is often not the most cost-effective in the long run.
Common Mistakes When Choosing an EDC System
Low-cost systems often lack scalability.
Complex systems reduce adoption.
Leads to data silos.
Creates regulatory risks.

Where Captivate Fits in 2026
Modern EDC platforms are evolving into integrated clinical ecosystems.
-
support modern clinical workflows
-
provide flexible study configuration
-
enable real-time data validation
-
scale across multiple studies
-
integrate with broader clinical systems
This makes them particularly relevant for:
-
CROs
-
biotech companies
-
modern research organizations
Decision Checklist (Quick Reference)
Before selecting an EDC system, ask:
-
Does it match trial complexity?
-
Is it compliant with regulations?
-
Is it easy for sites to use?
-
Can it integrate with other systems?
-
Can it scale with future trials?
-
Does the vendor provide strong support?