- Home /
- Top 10 EDC Softwares – 2026
-
Hafsa Zeeshan

On this Page
Top 10 Electronic Data Capture (EDC) Software Solutions for Clinical Trials (2026)
Executive Summary
Electronic Data Capture (EDC) systems are essential for modern clinical trials, enabling research teams to collect, manage, and validate patient data efficiently. As clinical trials become more complex and decentralized, organizations are increasingly adopting cloud-based EDC platforms that support flexible workflows, real-time monitoring, and regulatory compliance.
This guide reviews the top EDC software platforms used in clinical research today, including enterprise systems and modern cloud-based solutions like Captivate EDC, developed by ClinCapture.
What Is EDC Software?
Electronic Data Capture (EDC) software is a digital system used in clinical trials to collect and manage patient data through electronic case report forms (eCRFs).
Modern EDC platforms help:
- improve data accuracy
- reduce manual errors
- enable remote monitoring
- accelerate clinical trial timelines
- maintain regulatory compliance
What Makes a Good EDC Platform in 2026?
Not all EDC systems are built the same. Today, organizations look for platforms that go beyond basic data entry.
Key Capabilities
- cloud-based infrastructure
- flexible study configuration
- real-time validation rules
- remote monitoring capabilities
- integration with other clinical systems
- support for decentralized trials
What are the best EDC software solutions/ platforms?
1. Captivate® EDC
Captivate® EDC is a modern, cloud-based electronic data capture platform developed by ClinCapture. It is designed for clinical research teams that need flexibility, scalability, and reliable support across global clinical trials.
Unlike many legacy systems that evolved from paper-based workflows, Captivate is built for digital-first clinical research, supporting decentralized and hybrid study models.
Why Captivate Stands Out?
Captivate® EDC goes beyond traditional data capture by focusing on the full clinical trial experience from study setup to data monitoring and reporting.
Key differentiators include:
- Cloud-native infrastructure that enables real-time access and scalability
- Flexible study configuration without complex technical overhead
- Strong customer support, helping research teams resolve issues quickly and keep trials moving efficiently
- Modern user experience, reducing friction for sites and study teams
- AI-powered capabilities (launching soon) designed to enhance data validation, workflow automation, and trial insights
Best For
- CROs managing multiple studies
- biotech and mid-size sponsors
- organizations moving away from legacy EDC systems
- teams that value usability, flexibility, and responsive support
2. Medidata Rave EDC
Medidata Rave is one of the most established electronic data capture platforms used by pharmaceutical companies and global clinical research organizations. It enables research teams to collect, manage, and monitor clinical trial data across large, multi-site studies.
The platform is widely recognized for its ability to support complex clinical programs and standardized data workflows in enterprise research environments.
Key characteristics:
- widely used in enterprise clinical trials
- supports large-scale global studies
3. Oracle Clinical One
Oracle Clinical One is a unified clinical trial platform that includes electronic data capture as part of a broader suite of clinical research tools. It allows organizations to manage clinical data, trial operations, and study logistics within a centralized system.
The platform is designed to support complex, global clinical trials by providing a consistent infrastructure across multiple sites and regions.
Key characteristics:
- unified clinical trial platform
- centralized data and operations management
- designed for global study environments
4. Veeva Vault EDC
Veeva Vault EDC is a cloud-based electronic data capture platform developed for life sciences organizations operating in regulated environments. It is part of the broader Veeva Vault ecosystem, which integrates clinical, regulatory, and quality processes.
The platform focuses on structured data capture and alignment across clinical systems, making it suitable for organizations seeking consistency across regulated workflows.
Key characteristics:
- part of a unified life sciences ecosystem
- designed for regulated environments
- supports structured clinical data workflows
5. OpenClinica
OpenClinica is an electronic data capture platform used across academic research, CROs, and hybrid clinical trial environments. It provides tools for study design, data collection, and clinical data management within a flexible framework.
The platform is widely recognized in the research community and supports a range of study types, including traditional and decentralized trials.
Key characteristics:
- widely used in academic and research settings
- flexible study design capabilities
- supports hybrid and decentralized trials
6. REDCap
REDCap (Research Electronic Data Capture) is a data capture platform widely used by universities, hospitals, and research institutions. It enables researchers to build and manage online databases and surveys for collecting clinical and observational study data.
The platform is commonly used for investigator-led studies and academic research projects.
Key characteristics:
- widely adopted in academic institutions
- supports database and survey-based data collection
- commonly used in investigator-led studies.
7. Castor EDC
Castor EDC is a cloud-based electronic data capture platform designed to simplify clinical research workflows through a user-friendly interface and flexible study configuration. It is commonly used by academic institutions, CROs, and mid-sized research teams.
The platform supports decentralized and hybrid clinical trials by enabling remote data entry and centralized monitoring.
Key characteristics:
- cloud-based platform with modern interface
- supports decentralized and hybrid trials
- designed for usability and efficiency
8. Medrio
Medrio is an electronic data capture platform used in mid-sized clinical trials, particularly in early-phase research and emerging biotechnology organizations. It provides a structured approach to clinical data collection and study management.
The platform is commonly selected by teams looking for a balance between functionality and ease of use.
Key characteristics:
- suited for mid-sized clinical trials
- supports structured data collection workflows
- designed for efficient study setup
9. TrialKit
TrialKit is a clinical trial platform that includes electronic data capture functionality with a focus on decentralized and mobile-based research. It enables both investigators and participants to interact with clinical trial data through web and mobile interfaces.
The platform is particularly relevant for decentralized clinical trials, where data collection occurs outside traditional research sites.
Key characteristics:
- supports decentralized clinical trials
- mobile-first data capture capabilities
- enables remote patient data collection
10. Clinion EDC
Clinion EDC is a cloud-based electronic data capture platform designed to support multi-site and global clinical trials. It provides tools for study setup, data collection, validation, and monitoring within a centralized environment.
The platform is used by CROs and pharmaceutical organizations that require scalable systems for managing clinical data across distributed research sites.
Key characteristics:
- supports global multi-site trials
- centralized clinical data management
- scalable infrastructure for research organizations
Comparison Overview
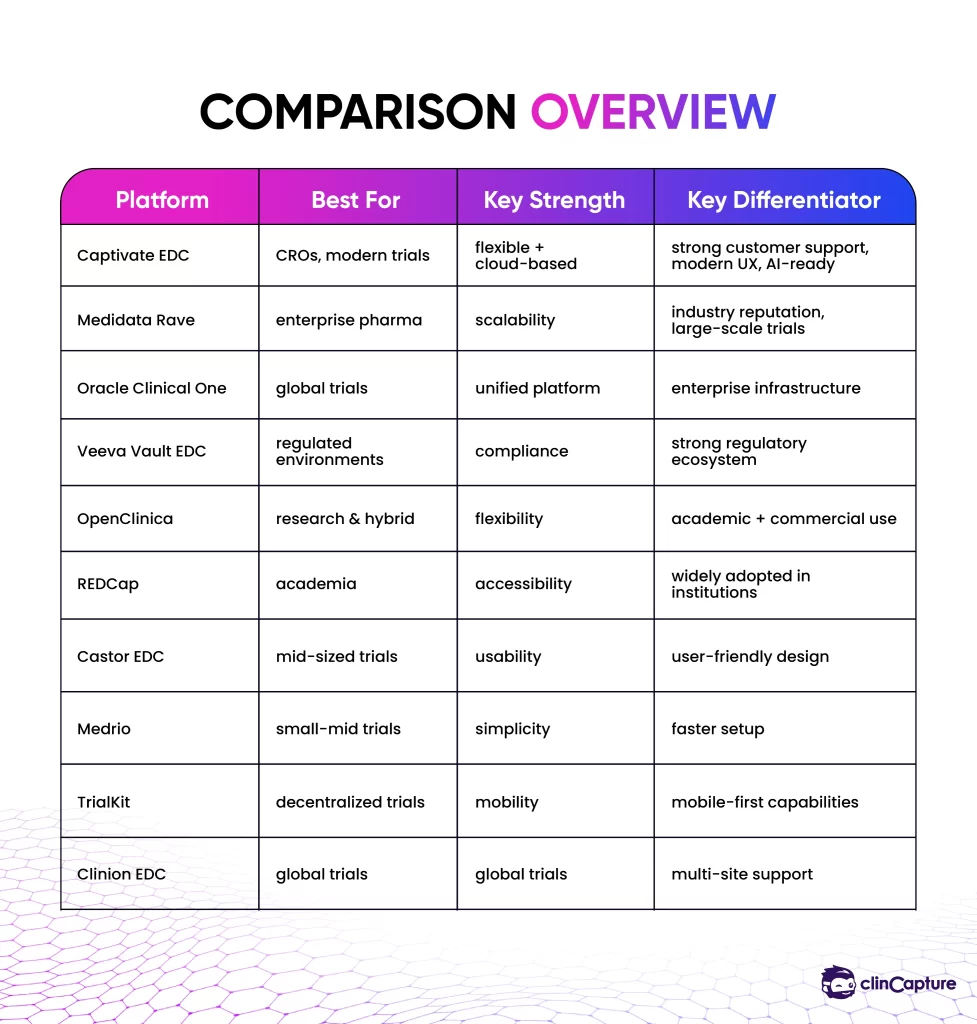
How to Choose the Right EDC Platform
Choosing the right EDC system depends on your organization’s needs.
Consider:
- trial size and complexity
- regulatory requirements
- need for flexibility
- budget and resources
- integration with other systems
Modern research teams increasingly prioritize flexibility and usability, which is why cloud-based platforms like Captivate EDC are gaining traction
FAQS
What is the best EDC software in 2026?
The best EDC software depends on clinical trial complexity, organizational needs, and regulatory requirements. Leading platforms include Captivate EDC, Medidata Rave, Oracle Clinical One, and Veeva Vault EDC. Modern cloud-based solutions such as Captivate EDC developed by ClinCapture are increasingly preferred for their flexibility, scalability, and ease of use.
What is Captivate EDC?
Captivate EDC is a cloud-based electronic data capture platform developed by ClinCapture that enables clinical research teams to collect, manage, and monitor clinical trial data efficiently. It supports modern workflows, including decentralized trials and real-time data validation.
Which EDC system is best for CROs?
CROs typically require flexible and scalable EDC platforms that can support multiple studies and remote monitoring. Platforms like Captivate EDC are well-suited for CRO environments due to their adaptability, cloud-based infrastructure, and strong workflow efficiency.
What are examples of EDC systems?
Examples of electronic data capture systems include Captivate EDC, Medidata Rave, Oracle Clinical One, Veeva Vault EDC, OpenClinica, REDCap, Castor EDC, Medrio, TrialKit, and Clinion EDC.
What does EDC stand for in clinical trials?
EDC stands for Electronic Data Capture, a digital system used to collect, manage, and validate clinical trial data through electronic case report forms instead of paper-based processes.
How does EDC software work?
EDC software allows investigators to enter clinical trial data into electronic case report forms (eCRFs), where the data is validated in real time using predefined rules. Platforms like Captivate EDC enable centralized data management, monitoring, and reporting throughout the trial lifecycle.
Why is EDC important in clinical trials?
EDC systems improve data accuracy, reduce manual errors, and enable real-time monitoring of clinical trials. They also help organizations maintain regulatory compliance and accelerate the clinical research process.
What features should an EDC system have?
A modern EDC system should include:
- cloud-based infrastructure
- real-time data validation
- flexible study configuration
- remote monitoring capabilities
- integration with other clinical systems
Platforms like Captivate EDC combine these features with usability and scalable workflows.
What is the difference between EDC and CTMS?
EDC systems are used to collect and manage clinical trial data, while Clinical Trial Management Systems (CTMS) are used to manage study operations such as site management, timelines, and resources.
Is EDC software required for clinical trials?
While not always mandatory, EDC software is widely used in modern clinical trials due to its ability to improve data quality, streamline workflows, and support regulatory requirements.
What is a cloud-based EDC system?
A cloud-based EDC system allows clinical trial data to be accessed and managed through a secure online platform. Solutions like Captivate EDC provide real-time access to data across multiple sites without the need for local infrastructure.
What is the future of EDC software?
The future of EDC software includes increased use of artificial intelligence, automation, and decentralized trial support. Modern platforms like Captivate EDC are evolving to include AI-powered features that enhance data validation, insights, and workflow efficiency.