-
ClinCapture

On this Page
What Is EDC (Electronic Data Capture) in Clinical Trials?
A Complete Research Guide to Electronic Data Capture Systems
Executive Summary
Electronic Data Capture (EDC) systems are digital platforms used in clinical research to collect, manage, validate, and store patient data during clinical trials. By replacing traditional paper-based case report forms, EDC systems enable real-time data entry, automated validation, remote monitoring, and regulatory compliance.
These capabilities significantly improve data accuracy, reduce trial timelines, and support decentralized clinical research models.
Modern EDC platforms are widely used by pharmaceutical companies, biotechnology firms, contract research organizations (CROs), and academic research institutions to manage complex clinical trials across multiple geographic locations. Leading EDC solutions provide integrated modules for electronic case report forms (eCRFs), Audit trails, Query management, and data monitoring
As clinical research becomes increasingly global and data-driven, EDC technology has evolved into a foundational component of modern clinical trial infrastructure. Cloud-based platforms such as Captivate® EDC and other enterprise clinical data systems now support scalable, secure, and regulatory-compliant research environments that accelerate drug development and improve trial efficiency.
Introduction: The Digital Transformation of Clinical Trials
Clinical trials are one of the most critical stages in the development of new medical treatments, pharmaceuticals, and medical devices. During these studies, researchers collect extensive patient data to evaluate the safety, efficacy, and regulatory compliance of investigational therapies
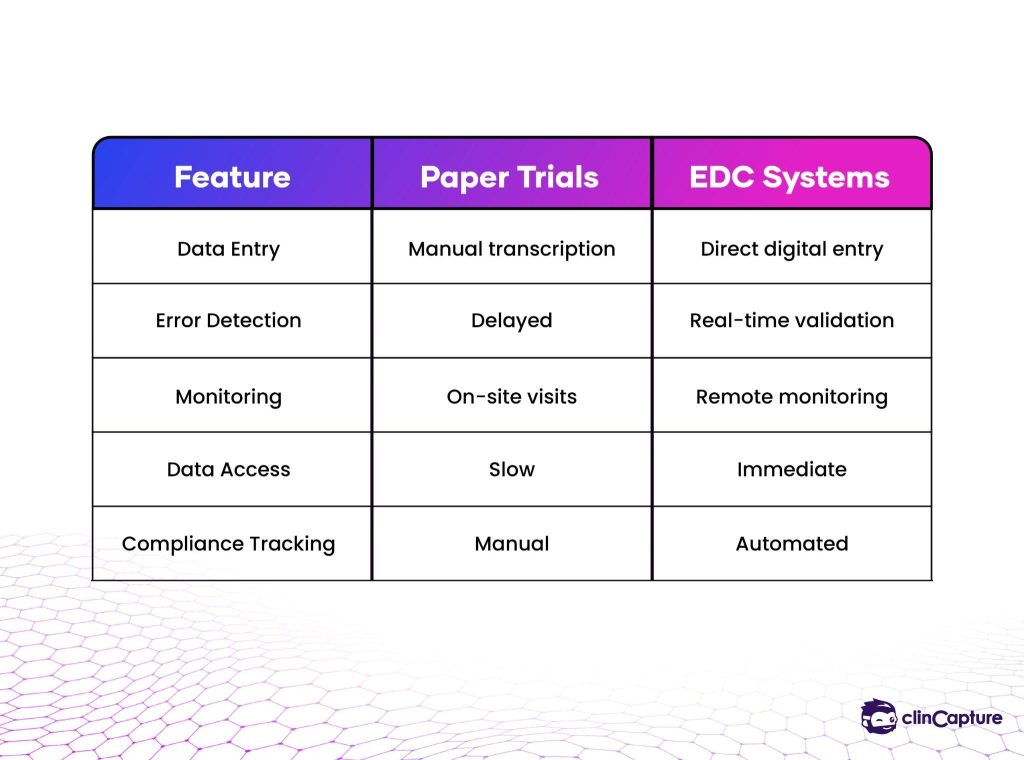
Historically, clinical trial data collection relied heavily on paper-based case report forms (CRFs).
Investigators manually recorded patient observations, laboratory results, and clinical measurements before submitting the documents for centralized data entry. This process was time-consuming, prone to transcription errors, and difficult to monitor in real time.
What Is Electronic Data Capture (EDC)?
Electronic Data Capture (EDC) refers to a digital system used in clinical trials to collect, manage, and store patient data through electronic case report forms (eCRFs). These systems replace paper-based clinical data collection and allow investigators to enter trial data directly into secure digital platforms.
- improving data accuracy
- reducing manual data entry errors
- enabling real-time monitoring of clinical trials
- ensuring regulatory compliance
- accelerating data analysis and reporting
The Evolution of Clinical Data Collection
Paper-Based Clinical Trials (Pre-2000)
- delayed data entry
- transcription errors
- limited oversight
- slow query resolution
Because data had to be manually entered into databases after collection, research teams often experienced significant delays between patient visits and data availability.
Early Electronic Data Capture Systems (2000–2010)
Cloud-Based EDC Platforms (2010–2020)
Modern Integrated eClinical Platforms (2020–Present)
Today, EDC platforms are often integrated into broader eClinical ecosystems that support additional clinical trial functions, including:
- electronic consent (eConsent)
- electronic patient-reported outcomes (ePRO)
- randomization and trial supply management (RTSM)
- Clinical trial analytics
- decentralized trial infrastructure
These integrated platforms help research organizations manage complex clinical trials using a unified digital environment.
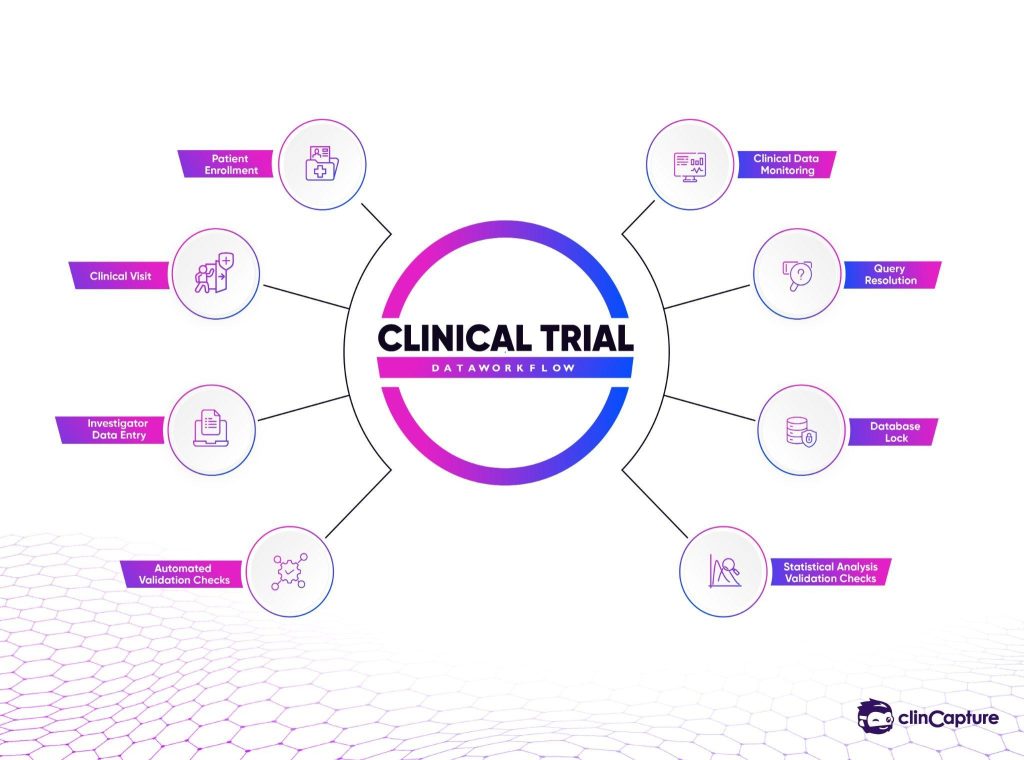
How EDC Systems Work in Clinical Trials
Electronic Data Capture systems operate through a structured workflow that supports the full lifecycle of clinical data collection.
Clinical Data Entry
Automated Data Validation
Modern EDC systems include automated validation rules that detect inconsistencies or missing information during data entry.
- range checks
- logical consistency checks
- mandatory field validation
- protocol compliance checks
Query Management
Data Monitoring
Database Lock and Analysis
Key Components of Modern EDC Platforms
Electronic Case Report Forms (eCRFs)
eCRFs are digital forms used to collect patient data during clinical trials. These forms are designed according to study protocols and regulatory requirements.
Validation Rules
Audit Trails
Role-Based Access Control
Data Integration
Benefits of EDC Systems in Clinical Research
Improved Data Accuracy
Faster Clinical Trials
Remote Monitoring
Clinical monitors can review trial data remotely, reducing travel costs and improving oversight
across multiple research sites.
Regulatory Compliance
EDC systems maintain detailed audit trails and security controls that support regulatory
compliance requirements such as 21 CFR Part 11.
Cost Efficiency
Digital data collection reduces administrative overhead and minimizes the need for manual data
transcription.
Adoption of EDC Across the Clinical Research Industry
Electronic Data Capture systems are now widely used across multiple sectors of the clinical research ecosystem.
Pharmaceutical Companies
Large pharmaceutical companies rely on EDC platforms to manage complex multi-site trials involving thousands of participants.
Biotechnology Firms
Biotech startups increasingly adopt cloud-based EDC platforms to manage early-phase trials with limited internal infrastructure.
Contract Research Organizations (CROs)
CROs frequently use EDC platforms to coordinate clinical trials on behalf of pharmaceutical sponsors.
Academic Research Institutions
Universities and research hospitals also rely on EDC systems to manage investigator-initiated clinical studies.
Adoption of EDC Across the Clinical Research Industry
Several software vendors provide EDC solutions for clinical trials.
Examples include:
These platforms vary in terms of scalability, user interface, and integration capabilities.
Modern platforms such as Captivate® EDC provide modular architectures that support broader clinical research workflows, including decentralized trials and electronic patient reporting.
How to Choose an EDC System
Selecting the appropriate EDC platform is a critical decision for research organizations.
Key evaluation criteria include:
- regulatory compliance features
- scalability for multi-site trials
- system usability for investigators
- integration with other clinical technologies
- cost and deployment models
Organizations must also consider long-term platform flexibility as clinical research methodologies continue evolving.
The Future of Electronic Data Capture
The next generation of EDC systems is expected to incorporate emerging technologies such as artificial intelligence, machine learning, and real-world data integration.
Future developments may include:
- automated data quality monitoring
- predictive analytics for trial outcomes
- decentralized patient data collection
- AI-assisted data validation
These innovations have the potential to further streamline clinical trial operations and improve the efficiency of medical research.
Conclusion
Electronic Data Capture systems have fundamentally transformed the way clinical trials collect and manage patient data. By replacing paper-based data collection with secure digital platforms, EDC technology improves data accuracy, accelerates research timelines, and enables remote monitoring of global clinical trials.
As clinical research becomes increasingly data-intensive, EDC systems will continue serving as a central component of the modern clinical trial ecosystem. Organizations that adopt advanced digital data management platforms will be better positioned to conduct efficient, compliant, and scalable clinical studies in the evolving landscape of medical research.
FAQS
What does EDC stand for in clinical trials?
EDC stands for Electronic Data Capture, a digital system used to collect and manage clinical trial data.
What is electronic data capture software?
Electronic data capture software is a platform used by clinical researchers to enter, validate, and manage patient data digitally.
Why is EDC used in clinical trials?
EDC systems improve data accuracy, enable real-time monitoring, and ensure regulatory compliance.
What are examples of EDC systems?
Examples include Captivate® EDC, Medidata Rave, Oracle Clinical One, OpenClinica, Castor EDC, and Captivate EDC.
How does EDC improve data accuracy?
EDC systems use automated validation rules that detect inconsistencies during data entry.
Is EDC required for clinical trials?
While not mandatory in all studies, EDC systems are widely used in modern clinical research due to their efficiency and compliance benefits.
What are electronic case report forms?
Electronic case report forms (eCRFs) are digital forms used to collect patient data during clinical trials.
How much does EDC software cost?
EDC pricing varies depending on trial complexity, number of sites, and system capabilities. Captivate® EDC by Clincapture is an affordable cloud-based EDC software with enhanced AI features, designed to provide everything you need for modern clinical trials in 2026.
What is cloud-based EDC?
A cloud-based EDC is an Electronic Data Capture system hosted online, allowing clinical trial teams to securely collect, manage, and access study data from anywhere in real time. Captivate® EDC by Clincapture is a cloud-based EDC platform that combines AI-powered features, secure data management, and an easy-to-use interface to help streamline clinical trials in 2026.
What industries use EDC systems?
Pharmaceutical companies, CROs, biotechnology firms, and academic research institutions use EDC platforms.