AI in clinical trials is addressing the challenges of escalating data complexity and accelerated timelines. AI in clinical trials is being used to handle increasing complexity, larger volumes of data, and shorter development timelines. It enhances not only operational efficiency but also the underlying approach to study design and management, facilitating faster and more reliable insights for strategic decisions.
Introduction: Clinical Research at an Inflection Point
Clinical trials remain the definitive mechanism for validating the safety, efficacy, and real-world applicability of new therapies and medical devices. Yet despite decades of methodological refinement, the operational realities of clinical research have become increasingly strained. Trial complexity has risen sharply, timelines continue to extend, and costs have escalated to levels that challenge both innovation velocity and patient access.
One of the clearest indicators of this shift is data scale. While eligibility criteria for clinical trial participants have remained largely consistent over the past decade, the volume of data generated in Phase III trials has tripled, reaching approximately 3.6 million data points per study, compared to levels observed ten years ago. This expansion is driven by the proliferation of electronic health records (EHRs), wearables, imaging, genomic datasets, decentralized trial technologies, and real-world data sources. Traditional operational models were not designed to manage, analyze, or act upon data at this magnitude or velocity.
At the same time, persistent structural inefficiencies remain unresolved:
- Nearly 80% of trials fail to meet enrollment timelines
- Protocol amendments remain common, costly, and disruptive
- Site performance variability continues to undermine predictability
- Manual oversight dominates areas that demand continuous, real-time intelligence
Against this backdrop, AI in clinical trials is no longer experimental or aspirational. It is becoming an operational necessity.
Industry benchmarks increasingly demonstrate measurable impact:
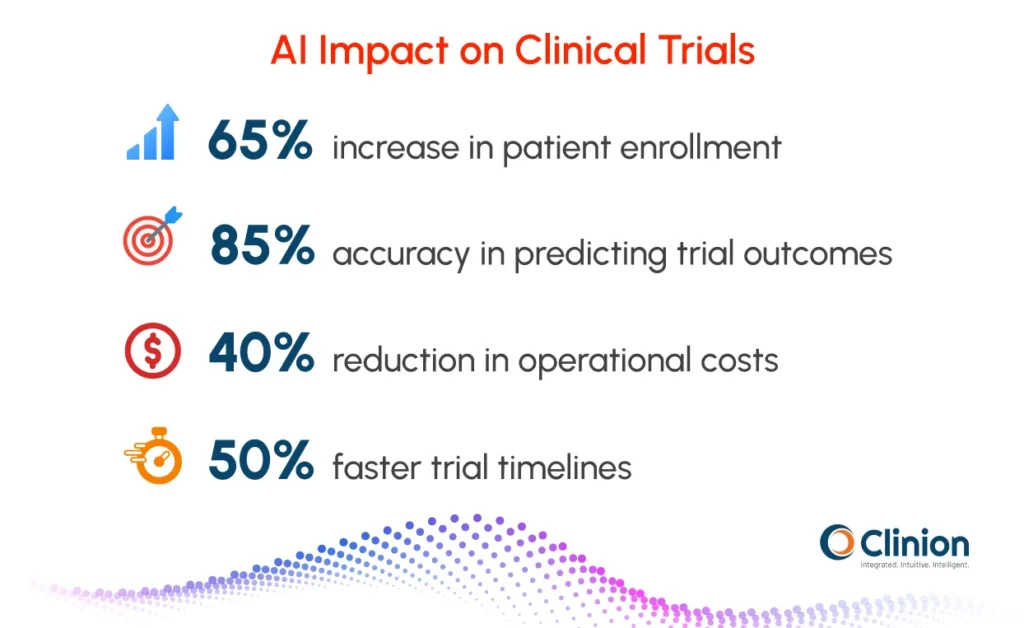
- AI-powered patient recruitment tools improve enrollment rates by up to 65%
- Predictive analytics models achieve approximately 85% accuracy in forecasting trial outcomes and site performance risks
- AI integration accelerates trial timelines by 30-50% while reducing operational costs by up to 40%
These gains are not the result of isolated tools, but of a broader shift toward clinical trial automation, where artificial intelligence and machine learning are embedded across the trial lifecycle.
This article examines how AI and machine learning in clinical trials are reshaping trial design, execution, monitoring, and evidence generation, while also addressing the regulatory, ethical, and operational constraints that govern their adoption.
Why Traditional Clinical Trial Models Are No Longer Sufficient
Clinical trials today operate in an environment characterized by:
- Multi-country, multi-site execution
- Increasingly narrow patient populations
- High protocol complexity
- Continuous data inflow from heterogeneous sources
Yet operational decision-making often relies on periodic reviews, static reports, and retrospective analysis. This disconnect between data availability and actionable insight is a fundamental limitation of legacy models.
Structural Inefficiencies Across the Trial Lifecycle
Key friction points persist across studies:
| Trial Phase | Persistent Challenges |
| Trial Design | Overly restrictive eligibility criteria, limited real-world relevance |
| Site Selection | Reliance on historical assumptions rather than predictive performance |
| Recruitment | Manual screening, low patient awareness, and high dropout rates |
| Trial Conduct | Reactive monitoring, delayed issue detection |
| Data Management | Manual cleaning, delayed locks, reconciliation bottlenecks |
| Reporting | Resource-intensive CSR development, long close-out cycles |
These inefficiencies compound over time, increasing both direct costs and opportunity costs.
The Role of AI in Clinical Research
Early adoption of automation in clinical trials focused on digitization – electronic data capture, electronic trial master files, and basic workflow automation. While valuable, these systems largely replicated manual processes in digital form.
In contrast, AI in clinical research introduces capabilities that are fundamentally different:
- Pattern recognition across high-dimensional datasets
- Probabilistic forecasting rather than rule-based triggers
- Continuous learning from historical and real-time data
- Decision support at scale
AI does not merely accelerate tasks; it alters how decisions are made.
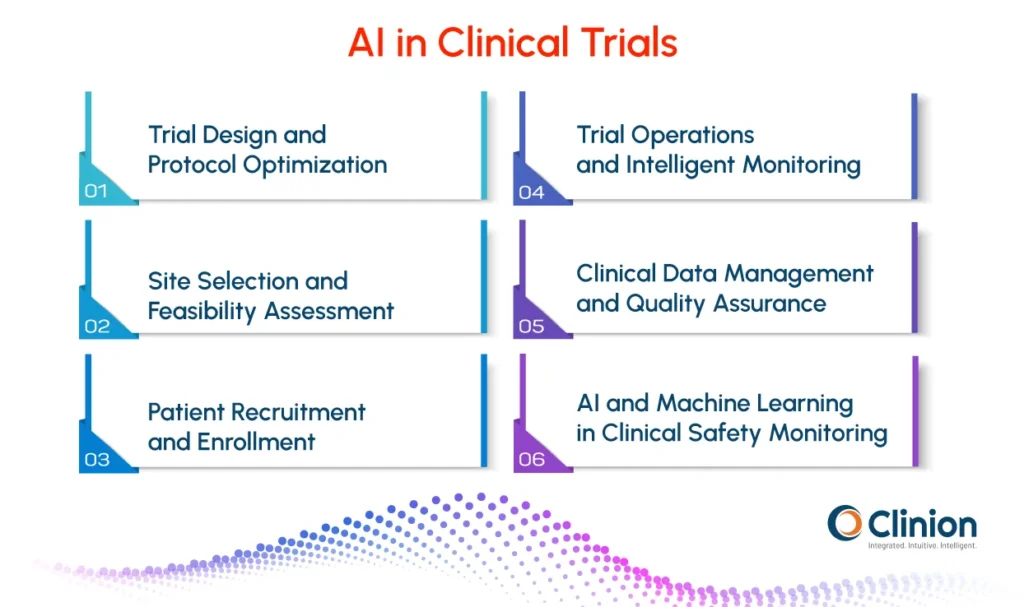
AI-Driven Trial Design and Protocol Optimization
Re-evaluating Eligibility Criteria Using Data
Eligibility criteria play a critical role in patient safety and scientific validity, yet they are often inherited from prior protocols without empirical reassessment. This contributes to unnecessarily narrow recruitment pools and reduced generalizability.
AI and machine learning models trained on historical trial and real-world datasets can:
- Identify criteria with minimal impact on safety or endpoints
- Quantify trade-offs between inclusivity and statistical power
- Simulate enrollment and outcome scenarios under modified criteria
Outcome: Broader, more representative cohorts without compromising trial integrity.
Adaptive and Predictive Trial Design
AI enables pre-trial simulation and ongoing optimization through:
- Predictive enrollment modeling
- Endpoint sensitivity analysis
- Scenario-based protocol feasibility testing
These capabilities reduce late-stage amendments and improve operational realism before first-patient-in.
AI in Site Selection and Feasibility Assessment
From Retrospective Metrics to Predictive Performance
Traditional site selection relies heavily on historical enrollment numbers and subjective feasibility questionnaires. AI-driven models expand this analysis by incorporating:
- Past enrollment velocity and screen failure rates
- Data entry timeliness and query resolution patterns
- Protocol deviation history
- Local patient population characteristics
- Competing trial density
Benefits of AI-Enabled Site Selection
- Reduced site over-activation
- Higher probability of on-time enrollment
- Early identification of underperforming sites
- More balanced geographic and demographic representation
This represents a shift from reactive site management to predictive site intelligence.
Regulatory Considerations for AI in Clinical Trials
egulatory authorities, including the FDA and EMA, increasingly acknowledge the role of AI in modernizing clinical research. However, acceptance is conditional. Rather than evaluating AI as a standalone innovation, regulators assess it through the lens of risk, accountability, and scientific validity.
Alignment with AI Governance Standards (ISO/IEC 42001)
As AI adoption matures, organizations are increasingly aligning their clinical AI systems with formal governance standards. ISO/IEC 42001, the international standard for AI management systems, provides a structured framework for governing AI across its lifecycle